Silver Tarnish Mystery: The Chemical Truth Behind Why It Turns Black

Table of Contents
- The Mysterious Transformation: When Beauty Turns Dark
- The Chemistry Unveiled: Silver Meets Sulfur
- Understanding Silver Sulfide Formation
- Environmental Culprits: Hidden Sources of Tarnish
- The Science of Tarnish Removal
- Prevention Strategies That Actually Work
- Modern Solutions and Ancient Wisdom
- Conclusion
- Frequently Asked Questions
The Mysterious Transformation: When Beauty Turns Dark
Sarah discovered her grandmother’s silver necklace buried in a jewelry box she hadn’t opened in five years. The piece that once gleamed with mirror-like brilliance now appeared as if it had been painted with charcoal. She held it up to the light, watching how even the deepest crevices had turned completely black, and wondered what mysterious force had transformed this precious metal so dramatically. The necklace wasn’t dirty in the traditional sense because no amount of soap and water would restore its original luster. Something far more profound had occurred, something that happened at the molecular level, something that revealed the hidden chemistry between silver and the invisible compounds floating in our atmosphere.
Proper maintenance requires understanding the difference between surface dirt and chemical transformation. Learning comprehensive care techniques for different precious metals helps jewelry owners develop appropriate cleaning routines that preserve rather than damage their valuable pieces through the years.
This transformation haunts everyone who owns silver jewelry, silverware, or decorative pieces. The phenomenon appears almost supernatural in its consistency and inevitability. You can store silver in the most careful conditions, wrap it in the softest cloth, place it in the darkest drawer, and still return months later to find that telltale darkness creeping across its surface. The tarnishing doesn’t discriminate between expensive heirloom pieces and affordable fashion jewelry, between freshly polished surfaces and those already showing their age. It’s a universal experience that connects jewelry owners across centuries and continents, from ancient civilizations who first worked with silver to modern collectors who invest thousands in precious metal pieces.
Before delving into the details of the article, watch this video which presents a scientific report on the chemical reactions that turn the shine of silver into a dull black layer :
The mystery deepens when you realize that other precious metals don’t behave this way. Gold maintains its shine indefinitely, platinum resists corrosion with remarkable stubbornness, and even copper develops that attractive green patina rather than an ugly black coating. Silver stands alone in its tendency to darken so completely and so quickly. This peculiarity has frustrated jewelry owners, challenged scientists, and created an entire industry devoted to silver care products. Understanding why silver behaves this way requires diving deep into chemistry, exploring the invisible world of atmospheric gases, and unraveling the complex dance of atoms and electrons that occurs on every silver surface exposed to air.
What makes silver tarnishing particularly frustrating is its seeming randomness. Two identical silver rings stored in the same drawer might tarnish at completely different rates. A silver bracelet worn every day might stay brighter than one carefully preserved in a jewelry box. Some people report their silver tarnishing within weeks, while others maintain shine for years under seemingly similar conditions. These inconsistencies have led to countless myths about silver care, from the belief that certain blood types cause faster tarnishing to ideas about magnetic fields affecting the process. The truth, as scientific research has revealed, is simultaneously more straightforward and more complex than any of these folk explanations.
The journey to understanding silver tarnish has occupied chemists and metallurgists for over a century. Early researchers noticed the phenomenon but lacked the tools to examine it at the molecular level. They observed that silver darkened in certain environments but not others, that some materials accelerated the process while others seemed protective, and that the blackness could be removed through various chemical treatments. However, the precise mechanisms remained elusive until modern analytical techniques allowed scientists to examine individual atoms and trace the exact pathways of chemical reactions. These discoveries have revolutionized how we understand, prevent, and reverse silver tarnishing.
Breakthrough computational research published in peer-reviewed chemistry journals revealed the atomic-level mechanisms of silver tarnish formation through sophisticated molecular dynamics simulations. These studies finally explained long-standing mysteries about why silver tarnishes so much faster than chemical theory predicted, opening new possibilities for developing truly tarnish-resistant silver materials for both decorative and technological applications.
Recent breakthroughs in computational chemistry have finally solved puzzles that stumped researchers for decades. Scientists using molecular dynamics simulations discovered that silver atoms don’t just sit passively waiting for sulfur to attack them. Instead, the silver itself actively participates in the tarnishing process in surprising ways. These findings explain why silver tarnishes so much faster than theory predicted and why tarnish layers grow thicker than the simple chemistry suggests they should. The implications extend far beyond jewelry care, affecting electronics manufacturing, historical artifact preservation, and even the development of new silver alloys that resist corrosion.
The Chemistry Unveiled: Silver Meets Sulfur
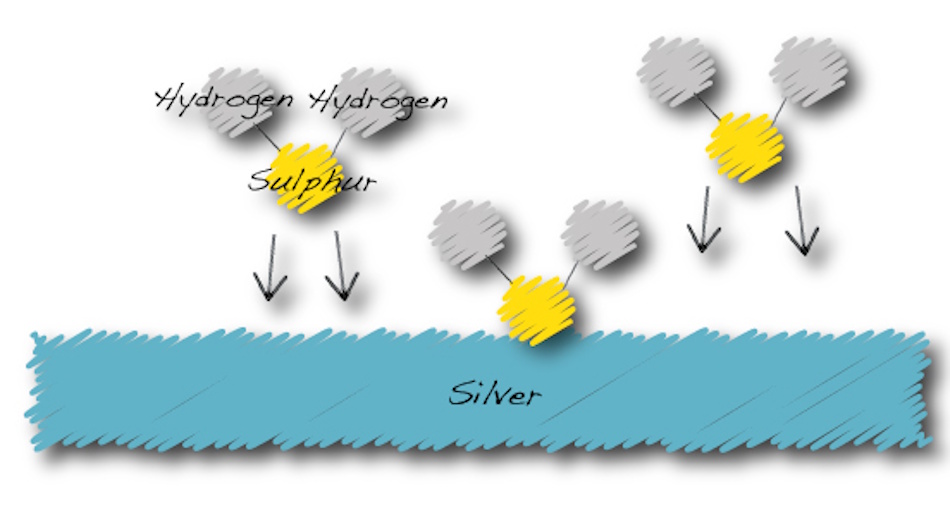
The blackening of silver represents one of the most common chemical transformations people witness in everyday life, yet few understand the precise molecular mechanisms driving this change. At its core, silver tarnishing involves a chemical reaction between silver atoms and sulfur-containing compounds present in the atmosphere. The primary culprit is hydrogen sulfide, a colorless gas with the notorious smell of rotten eggs, though in the concentrations that cause tarnishing, this gas remains completely undetectable to human senses. Even at concentrations as low as parts per billion, hydrogen sulfide can initiate the transformation of brilliant silver into dull black silver sulfide.
The chemical equation for this reaction appears deceptively simple. Silver atoms combine with hydrogen sulfide and oxygen to form silver sulfide, water, and release energy in the process. However, this straightforward equation masks an incredibly complex series of steps occurring at the atomic level. When hydrogen sulfide molecules approach a silver surface, they don’t simply bounce off or stick randomly. Instead, they interact with the silver’s crystal structure in specific ways, finding active sites where chemical bonds can form most easily. These active sites often exist at crystal boundaries, surface defects, or areas where the silver’s atomic arrangement differs slightly from its neighbors.
What happens next surprised researchers when they finally observed it through advanced simulation techniques. The sulfur atoms don’t merely attach to the surface silver atoms and stop there. Instead, once the initial sulfur atoms bond with surface silver, they create a sort of chemical vacuum that literally pulls silver atoms up from deeper layers. This upward migration of silver atoms through the developing sulfide layer explains why tarnish grows much thicker than simple surface chemistry would predict. The silver actively participates in its own tarnishing, moving toward sulfur atoms rather than waiting for sulfur to diffuse downward. This counter-intuitive behavior makes silver uniquely vulnerable to sulfidation compared to other metals.
The speed of this reaction depends on multiple factors working together in complex ways. Temperature plays a crucial role because chemical reactions generally proceed faster at higher temperatures, with the reaction rate roughly doubling for every ten-degree Celsius increase. Humidity provides the moisture necessary for ions to move freely, acting as an electrolyte that facilitates the electron transfer essential to the tarnishing reaction. Without adequate moisture, tarnishing proceeds at a glacial pace even in sulfur-rich environments. This explains why silver stored in extremely dry conditions like safety deposit boxes or climate-controlled vaults maintains its shine far longer than pieces kept in typical household environments.
The concentration of sulfur compounds matters enormously, but not in the linear way you might expect. Below certain threshold concentrations, tarnishing barely occurs because the silver’s natural oxide layer provides some protection. Above these thresholds, tarnishing accelerates dramatically. Environmental scientists have measured hydrogen sulfide concentrations in various settings, finding that urban areas with industrial pollution contain significantly higher levels than rural locations. Paper mills, petroleum refineries, geothermal areas, and locations near active volcanoes all show elevated hydrogen sulfide levels that accelerate silver tarnishing. Even wastewater treatment facilities and landfills contribute sulfur compounds to local atmospheres.
Federal environmental monitoring programs track atmospheric pollutants including sulfur compounds that affect not only human health but also materials degradation in indoor and outdoor environments. Understanding ambient hydrogen sulfide concentrations helps conservators and jewelry owners predict tarnishing rates and implement appropriate protective measures based on their specific geographic location and local pollution sources.
Oxygen’s role in silver tarnishing has been debated and researched extensively. While silver can technically form silver oxide when exposed to oxygen alone, this process occurs incredibly slowly and creates a nearly invisible layer. The oxide actually provides minor protection against further oxidation. However, when oxygen combines with sulfur compounds in the presence of moisture, it dramatically accelerates sulfide formation. The oxygen acts as a catalyst and reactant, participating directly in the chemical transformations while also creating conditions that favor rapid sulfidation. This synergistic effect explains why silver tarnishes so much faster in humid, sulfur-containing environments compared to dry ones.
Comprehensive chemical references detail silver’s fundamental properties including its reactivity with sulfur compounds and atmospheric gases. Understanding the element’s position in the periodic table and its electronic configuration helps explain why silver behaves differently from other precious metals when exposed to environmental conditions, providing the theoretical foundation for practical tarnish prevention strategies.
Understanding Silver Sulfide Formation
Silver sulfide, the black compound responsible for tarnish, possesses unique properties that distinguish it from both pure silver and other silver compounds. Chemically designated as Ag₂S, each molecule contains two silver atoms bonded to one sulfur atom. This specific stoichiometry creates a crystal structure fundamentally different from metallic silver’s arrangement. While pure silver atoms pack together in a face-centered cubic pattern that reflects light brilliantly, silver sulfide forms a different crystal system that absorbs most visible light wavelengths. This absorption characteristic gives tarnish its characteristic black appearance, though extremely thin layers can appear yellowish, golden, or even display rainbow iridescence before thickening into opaque black.
The formation of silver sulfide is thermodynamically favorable, meaning nature strongly prefers silver sulfide to exist rather than separate silver and sulfur. This explains why reversing tarnish requires active intervention rather than occurring spontaneously. The chemical bond between silver and sulfur is strong and stable, requiring significant energy input to break. When you examine the electron configurations involved, silver readily donates its valence electron while sulfur eagerly accepts electrons to complete its outer shell. This electron transfer creates ionic bonds supplemented by some covalent character, resulting in a stable compound that resists decomposition under normal conditions.
Academic chemistry resources provide detailed explanations of the electrochemical mechanisms involved in both tarnish formation and removal processes. These educational materials break down complex redox reactions into understandable steps, showing how electron transfers between aluminum and silver drive the cleaning reaction that jewelry owners observe when using the aluminum foil method.
Tarnish layers grow through a process called diffusion-limited growth. Initially, when clean silver first encounters sulfur compounds, surface atoms react quickly, creating a thin sulfide layer within hours or days depending on sulfur concentration. This initial layer forms unevenly, starting at the most reactive sites and gradually spreading across the entire surface. Once a continuous sulfide layer exists, further growth requires either sulfur atoms to diffuse inward through the sulfide to reach unreacted silver, or silver atoms to diffuse outward through the sulfide to meet incoming sulfur. Recent research revealed that silver diffusion outward dominates this process, contrary to earlier assumptions that sulfur diffusion inward was primary.
The thickness of tarnish layers varies depending on exposure duration and environmental conditions. In typical indoor environments, tarnish might reach thicknesses of several hundred nanometers over years. In highly polluted or sulfur-rich environments, the same thickness can develop in months or even weeks. Museums have documented cases where inadequate display case construction allowed sulfur-emitting materials to create severe tarnishing in just days. The relationship between thickness and appearance is nonlinear. Very thin layers create interference colors as light reflects from both the sulfide surface and the silver beneath, while thicker layers simply appear uniformly black.
Interestingly, silver sulfide formation is self-limiting to some degree. Once a thick sulfide layer develops, it provides a barrier that slows further sulfur penetration. The sulfide itself is relatively stable and doesn’t continue growing indefinitely without continued sulfur exposure. This protective quality means that tarnish, while visually unappealing, actually preserves the underlying silver from more aggressive forms of corrosion. Archaeological silver artifacts recovered from burial sites often show thick tarnish layers that protected the metal for centuries or even millennia. Without that tarnish, the silver might have corroded completely away.
The optical properties of silver sulfide explain why tarnish appears so dramatically dark. Silver sulfide has a bandgap that absorbs visible light efficiently, converting light energy into heat rather than reflecting it back to our eyes. This absorption occurs across the entire visible spectrum, making the sulfide appear black rather than colored. The crystal structure also matters because it scatters any light that manages to penetrate, further reducing reflectance. When you compare a tarnished silver surface to a polished one under a microscope, the difference is stark. The polished surface shows smooth, mirror-like reflectivity while the tarnished surface appears rough and light-absorbing at microscopic scales.
Environmental Culprits: Hidden Sources of Tarnish
The sulfur compounds that attack silver jewelry come from far more sources than most people realize. While industrial pollution and volcanic emissions contribute atmospheric hydrogen sulfide, everyday items in your home pose equal or greater threats to your silver. Eggs top the list of dangerous foods, containing sulfur-rich proteins that break down to release hydrogen sulfide when cooked or allowed to decay. The classic chemistry demonstration of intentionally tarnishing silver involves rubbing hard-boiled egg white on silver, which creates visible darkening in under an hour. This same mechanism explains why silverware stored near the kitchen, especially if used regularly for eggs, tarnishes faster than display pieces in other rooms.
Paper products deserve special attention as hidden tarnish accelerators. The kraft paper manufacturing process used for cardboard boxes, paper bags, and many packaging materials incorporates sulfur compounds. These compounds slowly release hydrogen sulfide vapors for years after manufacturing, creating a persistent source of tarnish-causing gas. Museum conservators have documented countless cases where silver artifacts stored in seemingly protective cardboard boxes developed severe tarnishing specifically from the box materials. Even gift boxes purchased from jewelry stores can contain kraft paper components that damage the very items they’re meant to protect. Acid-free, archival-quality papers exist specifically to avoid this problem, but typical consumer packaging rarely uses these more expensive materials.
Rubber products present another major threat that many silver owners don’t anticipate. Natural rubber contains vulcanizing agents and sulfur compounds added during manufacturing to improve elasticity and durability. Rubber bands, rubber grips on storage containers, rubber-backed carpets, and even the rubber seals on some jewelry boxes all emit sulfur vapors. The emission rate increases with temperature and age, meaning old rubber products become progressively more dangerous to nearby silver. Latex gloves, paradoxically often used for “protective” handling of silver, actually contain sulfur and can leave fingerprint-shaped tarnish patterns on silver surfaces.
Wool fabrics and some synthetic textiles also contribute to tarnish formation. Wool naturally contains sulfur-bearing amino acids like cysteine and methionine, which can break down and release hydrogen sulfide, especially in humid conditions. Storage of silver in wool-lined jewelry boxes, while seemingly luxurious and protective, can actually accelerate tarnishing unless the wool has been specially treated. Some synthetic fabrics treated with sulfur-containing dyes or finishes pose similar risks. Even cotton fabrics can be problematic if they’ve been treated with sulfur-based pesticides during cultivation or sulfur-containing fabric finishes during manufacturing.
Household cleaning products and personal care items frequently contain sulfur compounds that contribute to jewelry tarnishing. Certain shampoos, especially those designed for deep cleaning or dandruff treatment, contain sulfur-based ingredients. Hair permanent solutions and straightening treatments are particularly sulfur-rich. Body lotions, sunscreens, and makeup can all contain trace sulfur compounds. Chlorine bleach, while not sulfur-based, creates a harsh oxidizing environment that can accelerate tarnish formation through different chemical pathways. Swimming in chlorinated pools exposes silver jewelry to conditions that rapidly promote tarnishing and other forms of surface degradation.
The built environment itself can be a major source of tarnish-accelerating compounds. Certain paints, especially older oil-based formulations, release sulfur compounds as they cure and age. Foam insulation materials, adhesives, and sealants used in construction can emit volatile organic sulfur compounds for years. Wood products treated with certain preservatives become sulfur sources. Even concrete and mortar can contribute, particularly if they contain slag or other industrial byproducts. Buildings near industrial facilities, highways, or other pollution sources experience higher atmospheric sulfur levels that penetrate indoors through ventilation systems.
The Science of Tarnish Removal
Removing tarnish from silver can follow three fundamentally different approaches, each based on distinct chemical or physical principles. The most common method involves mechanical polishing, where abrasive particles physically scrape away the sulfide layer along with a thin layer of underlying silver. Commercial silver polishes typically contain fine particles of materials like calcium carbonate, alumina, or diatomaceous earth suspended in a cleaning solution. When you rub these polishes onto tarnished silver with a soft cloth, the abrasive particles scratch away microscopic amounts of material, gradually exposing fresh silver beneath. This method effectively restores shine but permanently removes silver atoms, slowly eroding fine details and reducing the weight of the object with each polishing.
Having proper cleaning supplies on hand enables regular maintenance before tarnish becomes severe and difficult to remove. Quality polishing cloths specifically designed for silver provide gentle cleaning action that removes developing tarnish without the harsh abrasion of coarser materials.
Shop on AliExpress via link: wholesale-silver-polishing-cloth
The abrasiveness of different polishing compounds varies enormously. Museum conservators use precipitated calcium carbonate, one of the gentlest abrasives available, to polish historically significant silver pieces. This compound has a Mohs hardness of only three, softer than most gemstones and many metal alloys, minimizing the risk of scratching while still removing tarnish through patient application. Commercial polishes intended for consumer use often employ more aggressive abrasives that work faster but remove more silver. Some products contain alumina particles with Mohs hardness approaching nine, capable of significant scratching if used carelessly. The particle size also matters enormously, with finer particles creating smoother finishes but requiring more effort to remove tarnish.
Chemical tarnish removal represents the second major approach, using solutions that chemically dissolve silver sulfide without mechanically scraping it away. Thiourea-based commercial dips work by forming a soluble complex with the silver sulfide, effectively pulling the sulfur away from the silver and allowing it to dissolve into the solution. These chemical dips work remarkably fast, often restoring shine within seconds to minutes. However, they come with significant drawbacks. Thiourea is classified as a probable carcinogen, requiring careful handling with protective equipment. The intensive washing required after treatment makes the method unsuitable for composite objects containing wood, ivory, or other materials that shouldn’t be soaked. The chemical action also leaves a characteristic micro-roughness on the silver surface that differs from the smooth polish achieved through mechanical methods.
Electrochemical tarnish removal, the third approach, has gained popularity among home users because it seems almost magical while requiring only simple household materials. The method involves placing tarnished silver in contact with aluminum foil in a solution of hot water containing baking soda or salt. This setup creates a galvanic cell where aluminum acts as the anode, silver as the cathode, and the solution as the electrolyte. Aluminum has a stronger tendency to donate electrons than silver, so it preferentially oxidizes while silver sulfide gets reduced back to metallic silver. The sulfide transfers from the silver to the aluminum, which gradually develops its own dark coating as the silver brightens.
The beauty of electrochemical cleaning lies in its selectivity. Unlike polishing, which removes both tarnish and silver indiscriminately, the electrochemical method specifically targets the sulfide layer while leaving the underlying silver largely intact. No silver is lost to abrasive removal, making the method appealing for maintaining the original surface and weight of valuable pieces. The reaction proceeds without harsh chemicals beyond baking soda and generates only aluminum sulfide as a byproduct, which poses no significant environmental or health risks. Watching the tarnish literally fade away before your eyes provides satisfying visual confirmation that the process works.
Educational chemistry demonstrations conducted by professional scientific organizations illustrate the electrochemical principles underlying this tarnish removal method. These accessible experiments help students and jewelry owners understand the fundamental redox reactions that occur during galvanic cleaning, demystifying what might otherwise seem like chemical magic into comprehensible science based on electron transfer between different metals.
However, electrochemical cleaning isn’t perfect for all applications. The method redistributes silver atoms across the surface in ways that create microscopic roughness over repeated treatments. Museum professionals generally avoid this technique for historically significant pieces because cumulative treatments gradually alter the original surface character. The method also works poorly on silver plated items where the thin silver layer has worn through in spots, and it’s completely unsuitable for composite objects that shouldn’t be submerged in water. Objects with sealed hollow spaces like candlesticks or teapots can trap solution inside, leading to ongoing chemical reactions that eventually cause damage from within.
Each tarnish removal method leaves different traces on the silver surface when examined under magnification. Mechanical polishing creates directional scratch patterns aligned with the polishing motion, with scratch depth depending on the abrasive particle size. Chemical dips produce a characteristically etched appearance with microscopic surface irregularities following the crystal structure of the silver. Electrochemical reduction creates a surface that looks smooth to the naked eye but shows microscopic redistribution of silver atoms under high magnification. These signature patterns allow conservation scientists to determine what cleaning methods have been used on historical artifacts, sometimes revealing problematic past treatments that require consideration when planning future conservation work.
Professional appraisal of silver pieces must account for cleaning history because different treatment methods leave distinct microscopic signatures that affect both condition assessment and value determination. Understanding how previous maintenance impacts current state helps collectors make informed decisions about appropriate care going forward and accurate valuation for insurance or sale purposes.
Prevention Strategies That Actually Work
Preventing tarnish proves far more effective than repeatedly removing it, both for maintaining silver’s appearance and for preserving its structural integrity. The foundation of any prevention strategy involves controlling the storage environment to minimize exposure to sulfur compounds, moisture, and oxygen. Anti-tarnish cloths and papers represent the first line of defense, specifically designed to absorb sulfur compounds from the air before they reach silver surfaces. These materials are typically cotton fabric treated with silver particles or zinc compounds that preferentially react with airborne sulfides. When silver is wrapped in these treated fabrics and placed in sealed plastic bags, the tarnishing rate drops dramatically compared to storage in ordinary cloth or paper.
The sealing component is crucial because it limits the total amount of air and sulfur compounds that can contact the silver. A simple polyethylene zip-top bag, while not perfectly airtight, reduces air exchange sufficiently to make a meaningful difference. Museum-quality storage goes further, using archival polyethylene bags with thicker walls and better seals. Some conservation specialists even evacuate air from storage bags or replace it with inert gases like argon or nitrogen, creating an environment where tarnishing simply cannot occur. These methods remain practical mainly for highly valuable pieces because they require specialized equipment and careful handling to maintain the oxygen-free environment.
Humidity control plays an enormous role in tarnish prevention because moisture facilitates the electrochemical reactions necessary for sulfide formation. Relative humidity above fifty percent allows a thin water layer to form on metal surfaces, creating the electrolyte conditions that enable ionic reactions to proceed rapidly. Below forty percent relative humidity, tarnish formation slows dramatically even in the presence of sulfur compounds. Silica gel packets, commonly found in product packaging, absorb atmospheric moisture and maintain lower humidity within sealed containers. Color-changing silica gel varieties provide visual confirmation when they’ve absorbed maximum moisture and need replacement or regeneration in an oven.
Implementing effective storage solutions requires having the right materials readily available. Organizing your silver pieces in appropriate compartments with proper humidity control materials keeps everything accessible while providing maximum protection against environmental factors that cause tarnishing.
Shop on AliExpress via link: wholesale-silver-jewelry-storage
Storage location within your home matters more than most people realize. Bathrooms, kitchens, and basements typically experience higher humidity and temperature fluctuations, accelerating tarnishing. Bedrooms and living areas usually provide more stable conditions. Climate-controlled spaces with consistent temperature and humidity offer ideal storage conditions. Attics should generally be avoided due to extreme temperature swings and often high humidity. The specific materials surrounding stored silver also require attention. Wooden jewelry boxes, while attractive, can emit acetic acid and other volatile compounds that accelerate corrosion. Sulfur-free tissue paper costs more than standard paper but pays off in reduced tarnishing. Foam padding and rubber gaskets should be avoided in favor of untreated cotton or polyester batting.
Active use of silver jewelry provides surprising protection against tarnishing. Body oils create a thin organic coating that provides a barrier between silver and atmospheric sulfides. The mechanical action of wearing jewelry constantly removes the very thin initial tarnish layers before they become visible. This explains the common observation that frequently worn rings and necklaces stay brighter than occasionally worn pieces. Professional jewelers often advise customers to rotate their silver pieces regularly rather than keeping favorites stored away. The adage “use it or lose it” applies perfectly to silver maintenance.
Protective coatings offer another prevention avenue, though they come with tradeoffs. Clear lacquer coatings seal silver from atmospheric exposure, preventing tarnish formation as long as the coating remains intact. However, lacquer eventually scratches, yellows, and degrades, requiring professional removal and reapplication. Rhodium plating, commonly applied to white gold jewelry, works exceptionally well for silver, providing a hard, bright, tarnish-resistant surface. The rhodium layer typically lasts one to three years with regular wear before requiring reapplication. Some owners love the maintenance-free shine that plating provides, while others prefer maintaining uncoated silver to preserve its original surface character and unique warm luster.
Different approaches to jewelry maintenance reflect personal preferences about balancing convenience with authenticity. Statement pieces that serve as focal points in jewelry collections often receive more careful preservation attention because their visual impact depends on maintaining pristine condition and distinctive appearance that sets them apart from everyday accessories.
Modern technology has introduced new prevention options that would have seemed like science fiction decades ago. Atomic layer deposition can apply coating layers just nanometers thick, providing protection while remaining virtually invisible and preserving the tactile and visual qualities of the silver surface. These coatings can be designed to be reversible, addressing one of the main concerns conservators have with permanent protective treatments. Nanomaterial coatings incorporating zinc oxide or other reactive compounds can scavenge sulfur compounds before they reach the silver surface. While these technologies currently remain expensive and mainly available through specialized conservation laboratories, they represent the future of silver protection for valuable pieces.
Modern Solutions and Ancient Wisdom
The contrast between ancient silver care practices and modern scientific approaches reveals fascinating continuities alongside dramatic innovations. Ancient civilizations that worked extensively with silver developed empirical knowledge about tarnish removal long before chemistry existed as a science. Roman households used a paste of chalk and vinegar to polish silver, effectively creating a mild acid solution with an abrasive component. Medieval Europeans discovered that burying tarnished silver in wood ash then rinsing it clean restored shine through the alkaline properties of the ash. These traditional methods worked through the same chemical and physical principles we understand today, even though their practitioners couldn’t explain why they succeeded.
Victorian-era servants spent countless hours polishing the vast quantities of silver found in wealthy households, developing techniques and products that remain relevant today. The connection between egg and tarnish was already well-known, leading to careful washing of silver utensils immediately after meals featuring eggs. The practice of storing silver in flannel bags traces back centuries, with the fabric providing both cushioning and minor tarnish inhibition. Some Victorian polishing recipes called for jeweler’s rouge, essentially iron oxide particles that provided gentle abrasion. Others used whiting, pulverized chalk that achieved similar results. These historical practices demonstrate sophisticated practical knowledge gained through observation and experimentation across generations.
Historical silver pieces from earlier eras often display characteristic wear patterns that reveal their usage history and cleaning practices over decades or centuries. [Collectors seeking vintage silver jewelry ](https://infoprods.com/blog/vintage-jewelry-treasure-huntingCollectors seeking vintage silver jewelry)should understand these aging signatures to distinguish authentic patina from damage and to appreciate the stories these pieces tell through their physical condition.
The scientific revolution in silver care began in the early twentieth century when chemists finally understood the molecular basis of tarnishing. Research conducted by the United States Bureau of Standards and Bureau of Mines in the nineteen twenties systematically investigated silver alloys and their tarnish resistance. These studies established that adding small amounts of certain metals could slow tarnishing, though usually at the cost of other desirable properties like softness and malleability. The development of stainless steel and other corrosion-resistant alloys inspired similar efforts with silver, though no completely tarnish-proof silver alloy has ever been commercialized successfully because modifications that resist tarnish typically compromise other essential characteristics.
Scientists from government research institutions have conducted extensive metallurgical studies examining how different alloying elements affect silver’s chemical properties and corrosion resistance. The pioneering work established fundamental principles that continue guiding modern silver alloy development, though achieving perfect tarnish resistance while maintaining all of silver’s desirable characteristics remains an ongoing challenge in materials science.
Conservation science emerged as a distinct discipline in the mid-twentieth century, bringing rigorous scientific methods to the preservation of cultural heritage. Museums realized that their traditional silver cleaning practices, while producing beautiful results, gradually destroyed the very objects they sought to preserve. This recognition led to development of gentler cleaning methods using the mildest possible abrasives and careful technique. Conservation scientists began testing materials to identify which papers, adhesives, fabrics, and display case components were safe for silver storage versus which would accelerate degradation. Their findings revolutionized museum practice and gradually filtered into public knowledge, though many dangerous materials remain in common use simply because alternatives cost more.
National conservation agencies have developed comprehensive technical guidelines for proper silver storage and maintenance based on decades of scientific research and practical experience with museum collections. These authoritative resources provide detailed specifications for materials selection, environmental control, and cleaning methods that balance preservation needs with practical accessibility for both institutions and private collectors.
The twenty-first century brought computational chemistry and nanotechnology into the silver preservation arena. Researchers at institutions like Trinity College Dublin and Nokia Bell Labs used molecular dynamics simulations to watch individual atoms during tarnish formation, finally solving mysteries that had persisted for over a century. These simulations revealed that silver atoms actively migrate toward sulfur during sulfide formation, a counter-intuitive behavior that explains why tarnish grows thicker than simple surface chemistry predicts. Armed with this knowledge, materials scientists can now design silver alloys with dopant atoms that inhibit silver diffusion, potentially creating truly tarnish-resistant silver for electronics and other applications where surface conductivity matters critically.
Nanotechnology applications to silver conservation represent cutting-edge research with profound implications. Scientists at the University of Maryland and the Walters Art Museum are developing atomic layer deposition techniques to apply protective coatings just nanometers thick to silver artifacts. These ultra-thin coatings provide protection while remaining essentially invisible and preservible key aesthetic qualities. Most importantly, they can be designed for complete reversibility, allowing future conservators to remove the coating and apply more advanced treatments as technology progresses. This reversibility addresses a fundamental principle of modern conservation ethics: interventions should be reversible because future generations will develop better methods we cannot yet imagine.
Conclusion
The blackening of silver represents one of chemistry’s most visible demonstrations, a transformation that occurs in homes worldwide every single day. What appears as simple darkening actually involves complex molecular mechanisms where silver atoms migrate through growing sulfide layers to meet incoming sulfur compounds. The reaction’s inevitability stems from thermodynamic favorability, with nature strongly preferring the stable silver sulfide compound over separated elements. Understanding this chemistry transforms tarnish from a mysterious curse into an understandable, manageable phenomenon.
Prevention strategies based on controlling environmental factors work far better than endless cycles of cleaning. Limiting exposure to sulfur compounds, controlling humidity, using proper storage materials, and even regular wear of silver jewelry all dramatically reduce tarnishing rates. When tarnish does develop, choosing appropriate removal methods based on the object’s value and characteristics preserves silver for future generations. The ongoing development of new protective coatings and tarnish-resistant alloys promises to make silver care easier while maintaining the unique beauty that has made silver precious throughout human history. Your grandmother’s blackened necklace can shine again, and with proper care, it will pass to future generations still gleaming with the warm luster that only silver provides.
Frequently Asked Questions
Question 1: Why does silver turn black instead of other colors?
Answer 1: Silver turns black specifically because it forms silver sulfide when exposed to hydrogen sulfide in the air. This particular chemical compound, with the formula Ag₂S, has a crystal structure and electronic configuration that absorbs light across the entire visible spectrum rather than reflecting it back to our eyes. When light hits silver sulfide, the energy is converted to heat rather than being reflected as it would be from pure silver. Other metals form different compounds with different optical properties when they corrode, which is why copper turns green from copper carbonate formation, iron turns reddish-brown from iron oxide, and aluminum develops a grayish-white oxide layer. The specific black color of tarnished silver is an inherent property of the silver sulfide molecule and cannot be changed without altering the chemical composition. Even extremely thin layers of silver sulfide appear yellowish or golden before thickening to black, demonstrating how the optical properties depend on layer thickness and light interference effects at the nanometer scale.
Question 2: Can tarnish damage my silver jewelry permanently?
Answer 2: Tarnish itself does not cause permanent damage to silver because it forms only a thin surface layer that actually protects the underlying metal from further corrosion. The silver sulfide that creates the black appearance is chemically stable and self-limiting, meaning it stops growing once it reaches a certain thickness. However, the real damage comes from repeated aggressive cleaning to remove tarnish. Each time you polish silver with abrasive compounds, you remove not just the tarnish but also a microscopic layer of the actual silver beneath it. Over many years of repeated polishing, this gradual material loss becomes visible as the wearing away of fine details, the rounding of sharp edges, and the fading of engraved decorations. Historic silver pieces that have been polished for centuries often show significant detail loss compared to their original condition. Museum conservators take great care to minimize cleaning frequency and use the gentlest possible methods precisely because they understand that the cure can be worse than the disease. The key to preserving silver long-term involves preventing tarnish through proper storage rather than constantly removing it through aggressive polishing.
Question 3: Does pure silver tarnish faster than sterling silver?
Answer 3: Pure silver, also called fine silver, actually tarnishes slightly slower than sterling silver under most conditions, though the difference is not dramatic in typical indoor environments. Sterling silver, which contains ninety-two point five percent silver and seven point five percent copper or other alloying metals, can tarnish somewhat faster because the copper component reacts with sulfur compounds and can form its own sulfide alongside the silver sulfide. The copper sulfide contributes additional darkening and can sometimes create a brownish tint rather than pure black. However, the alloying metals in sterling silver provide significant practical benefits that outweigh this minor disadvantage. Pure silver is extremely soft and malleable, making it unsuitable for jewelry and utensils that must withstand handling and use without bending or scratching easily. The copper or other metals added to create sterling silver dramatically increase hardness and durability while only marginally affecting tarnish resistance. In highly polluted environments or when exposed to specific chemicals that react preferentially with copper, the difference in tarnishing rates becomes more noticeable. Some specialized silver alloys incorporate elements like germanium or palladium that can provide improved tarnish resistance compared to traditional sterling, though these alloys cost more and may have different working properties that jewelers must accommodate during fabrication.
Question 4: What everyday items cause silver to tarnish quickly?
Answer 4: Many common household items accelerate silver tarnishing through the release of sulfur compounds, often in ways that surprise silver owners. Eggs rank among the most potent tarnish accelerators because they contain sulfur-rich proteins that break down to release hydrogen sulfide gas, particularly when cooked. Even handling silver after touching raw eggs can transfer enough sulfur compounds to create visible tarnish spots. Rubber products including rubber bands, rubber gaskets, latex gloves, and rubber-backed carpets all contain vulcanizing sulfur that slowly vaporizes over time. Wool fabrics naturally contain sulfur-bearing amino acids that can decompose and release hydrogen sulfide, especially in humid conditions. Many paper products, particularly cardboard boxes and kraft paper, are manufactured using sulfur compounds that continue releasing tarnish-causing vapors for years after production. Certain cosmetics, perfumes, hair permanents, and sunscreens contain sulfur-based ingredients that can tarnish silver jewelry through direct contact. Even some foods like onions, mustard, and certain pickled products contain sulfur compounds that can accelerate tarnishing. Household cleaners, particularly those containing chlorine bleach, create harsh oxidizing conditions that contribute to surface degradation beyond simple tarnishing. The cumulative effect of these various sources means that silver stored or used in typical household environments faces constant exposure to tarnish-causing compounds, explaining why even carefully stored pieces gradually darken over time.
Question 5: Is aluminum foil cleaning safe for valuable silver?
Answer 5: Aluminum foil cleaning using the electrochemical reduction method is generally safe for most silver items and actually causes less material loss than abrasive polishing. The method works by creating a galvanic cell where electrons transfer from aluminum to the silver sulfide, reducing it back to metallic silver without physically scraping away material. No silver is lost through abrasion, which makes the method appealing from a conservation standpoint. However, museum conservators and professional restorers typically avoid this method for historically significant pieces, valuable antiques, and rare coins despite its gentleness regarding material loss. The reason relates to how the method redistributes silver atoms across the surface. When sulfide converts back to silver, the atoms don’t necessarily return to their exact original positions in the silver crystal structure. Instead, they deposit somewhat randomly, creating microscopic surface roughness that accumulates with repeated treatments. Under high magnification, surfaces cleaned repeatedly with the electrochemical method show a characteristic texture different from original polished surfaces. For ordinary jewelry and silverware where historical authenticity matters less than appearance and function, the aluminum foil method provides an excellent option that’s safe, inexpensive, and effective. For truly valuable pieces, consultation with a professional conservator about appropriate cleaning methods specific to that particular object’s composition, condition, and historical significance makes more sense than do-it-yourself treatments regardless of how gentle they appear.
Question 6: Can silver tarnish in sealed containers?
Answer 6: Silver can definitely tarnish even in sealed containers if sulfur compounds are present inside the sealed environment or if the seal isn’t truly airtight. Many materials commonly used in jewelry boxes, storage containers, and packaging emit sulfur-containing vapors over time, creating a tarnishing atmosphere even within supposedly protective enclosures. Cardboard boxes, foam padding, rubber gaskets, certain adhesives, and wood products all can release hydrogen sulfide or other sulfur compounds that accumulate in sealed spaces and attack silver surfaces. The sealed environment can actually accelerate tarnishing in these situations because the sulfur compounds become concentrated rather than dispersing into the larger atmosphere. Moisture trapped inside sealed containers also facilitates tarnishing by providing the electrolyte environment necessary for the chemical reactions to proceed efficiently. The only truly effective sealed storage involves using containers made from inert materials like polyethylene plastic, lining them with anti-tarnish cloth or paper specifically designed to absorb sulfur compounds, adding silica gel packets to control moisture, and ensuring that no sulfur-emitting materials are present inside. Museum-quality storage systems may even evacuate air and backfill with inert gases like nitrogen or argon to eliminate oxygen and prevent any chemical reactions. Simply placing silver in any sealed container without considering the materials used in construction and the atmospheric composition inside provides no protection and may actually create worse conditions than open storage in many cases.
Question 7: Why does some silver tarnish appear rainbow-colored?
Answer 7: Rainbow or iridescent tarnish develops when silver sulfide layers are extremely thin, typically measured in nanometers, creating optical interference patterns similar to those seen in soap bubbles or oil films on water. When light strikes these ultra-thin layers, some reflects from the top surface of the sulfide while other light penetrates through to reflect from the silver beneath. These two reflected light waves interfere with each other either constructively or destructively depending on the sulfide layer thickness and the wavelength of the light. Different wavelengths experience different interference patterns at any given thickness, causing some colors to amplify while others cancel out. The result appears as vivid iridescent colors that shift as the viewing angle changes or as you move across the surface where thickness varies slightly. The specific color sequence as tarnish thickens progresses from no visible tarnish on perfectly clean silver, through pale yellow, to gold, then bronze, followed by purple and blue iridescence, and finally to the uniform black characteristic of thick tarnish. This color progression corresponds to increasing sulfide thickness from roughly ten nanometers for the first hints of color through several hundred nanometers for the iridescent blues, and beyond to opaque black when the layer exceeds about a thousand nanometers. Some silver owners actually appreciate the iridescent stage of tarnishing for its unique beauty, though it represents an intermediate degradation state that will inevitably progress to uniform black without intervention. Understanding this progression allows you to estimate approximately how long tarnish has been developing based on its appearance.
Question 8: Does humidity affect how fast silver tarnishes?
Answer 8: Humidity dramatically accelerates silver tarnishing because water molecules adsorbed onto the metal surface create a thin electrolyte layer that facilitates the chemical reactions necessary for sulfide formation. At relative humidity levels below forty percent, silver surfaces remain essentially dry at the molecular level and tarnishing proceeds very slowly even in the presence of sulfur compounds. As humidity rises above forty percent and approaches fifty percent, a continuous water monolayer begins forming on the silver surface. This water layer allows hydrogen sulfide molecules to dissolve and dissociate into hydrogen ions and sulfide ions, while also enabling silver atoms to ionize and move more freely. The presence of this aqueous environment transforms tarnishing from a slow solid-state reaction into a much faster electrochemical process where charged particles move readily and react efficiently. Above fifty percent relative humidity, multiple water layers form and reactions accelerate further. Museum conservation standards typically specify maintaining silver storage environments below forty percent relative humidity specifically because of this dramatic effect. Temperature interacts with humidity in complex ways because warm air holds more moisture than cool air at the same relative humidity percentage, and chemical reaction rates also increase with temperature independent of moisture effects. The combination of high temperature and high humidity creates the worst conditions for silver preservation, while cool, dry environments dramatically slow tarnishing. This explains why silver stored in climate-controlled museum vaults or safety deposit boxes in temperature and humidity regulated bank basements maintains its shine far longer than pieces kept in typical homes where humidity fluctuates seasonally and may spike very high during summer months or in bathrooms and kitchens.
Question 9: Can I prevent silver tarnish completely?
Answer 9: Complete prevention of silver tarnish is theoretically impossible as long as any sulfur compounds can reach the silver surface, but you can reduce tarnishing to negligible levels through proper storage and environmental control. The most effective approach combines multiple protective strategies: storing silver wrapped in anti-tarnish cloth or paper within sealed polyethylene bags, adding silica gel packets to control moisture, maintaining low humidity environments below forty percent relative humidity, using storage containers made from inert materials that don’t emit sulfur compounds, and keeping silver away from sulfur sources like rubber, wool, eggs, and certain paper products. Some owners take additional steps like storing silver in safety deposit boxes where temperature and humidity are controlled professionally, or even in inert gas environments where oxygen has been replaced with nitrogen or argon. Regular use of silver jewelry actually provides protection because body oils create a barrier and mechanical action removes developing tarnish before it becomes visible. Protective coatings represent another option, with rhodium plating providing excellent tarnish resistance for one to three years before requiring reapplication, or specialized lacquers sealing silver from atmospheric exposure though these eventually require professional removal and replacement. Modern nanotechnology approaches using atomic layer deposition can apply virtually invisible protective coatings just nanometers thick that may revolutionize silver preservation for valuable pieces. While none of these methods prevent tarnishing absolutely forever, combining them can extend the time between necessary cleanings from weeks or months to years or even decades, effectively making tarnish a non-issue for practical purposes while preserving the silver’s original surface and minimizing the material loss associated with repeated cleaning cycles.
Question 10: Why do some silver pieces tarnish while others don’t?
Answer 10: Different silver pieces tarnish at dramatically varying rates due to differences in alloy composition, surface finish, storage conditions, usage patterns, and local environmental factors. Sterling silver pieces containing more reactive alloying metals like copper may tarnish slightly faster than pure silver or pieces alloyed with less reactive metals. Surface finish matters because highly polished surfaces with fewer microscopic irregularities provide fewer active sites where tarnish reactions can initiate, while matte or textured surfaces offer more reactive surface area. Silver pieces worn regularly benefit from the protective barrier created by body oils and from mechanical action that removes developing tarnish continuously before it becomes visible, explaining why frequently worn rings stay brighter than identical pieces stored in drawers. Storage location creates enormous differences, with pieces kept in bathrooms or kitchens experiencing higher humidity and temperature fluctuations that accelerate tarnishing compared to pieces in climate-controlled bedrooms or living areas. Proximity to sulfur sources makes a huge difference, so silver stored near rubber bands, cardboard boxes, wool fabrics, or kitchen areas where eggs are frequently cooked will tarnish much faster than pieces kept in carefully selected storage away from these hazards. Silver-plated items tarnish differently than solid silver because the thin plating may wear through in high-friction areas, exposing base metal that oxidizes differently. Pieces with protective coatings like lacquer or rhodium plating don’t tarnish at all until the coating degrades or wears through. Even individual body chemistry variations affect tarnishing rates for worn jewelry, with some people’s perspiration composition accelerating tarnish while others seem to keep silver bright indefinitely. The combination of all these factors explains why two nominally identical silver pieces can show completely different tarnishing patterns, frustrating owners who wonder why their favorite necklace stays bright while another rarely worn one turns black.
Articles related:
Tags
📧 Get More Articles Like This
Subscribe to receive product reviews and buying guides in your inbox!
We respect your privacy. Unsubscribe at any time.