Non-Invasive Glucose Monitors: Smart Watches vs Patches vs Implants Battle

- The Promise of Pain-Free Glucose Monitoring
- Understanding Current Glucose Monitoring Technologies
- Smart Watch Integration: The Reality vs The Hype
- Patch-Based CGM Systems: The Current Gold Standard
- Implantable Sensors: Long-Term Solutions
- Accuracy Comparison: Which Technology Performs Best
- Cost Analysis and Insurance Coverage in 2026
- The Future: What’s Coming Next
- Making Your Choice: Decision Framework
- FAQ
The Promise of Pain-Free Glucose Monitoring
Sarah Martinez remembers the exact moment she stopped feeling her fingertips. After fourteen years of Type 1 diabetes and roughly fifteen thousand finger-prick tests, the constant nerve damage had finally caught up with her daily monitoring routine. Each morning began with the familiar ritual of loading a lancet, choosing a less-damaged spot on her fingers, and hoping the blood drop would be large enough for her glucose meter to register a reading. Sometimes it took three or four attempts before she could get enough blood for a successful test, and the accumulating scar tissue made each prick increasingly difficult. Her endocrinologist had been recommending continuous glucose monitoring for years, but Sarah resisted the idea of wearing visible medical devices that would announce her condition to everyone around her. That changed one morning when she couldn’t wake her teenage daughter for school after experiencing severe overnight hypoglycemia that her traditional monitoring completely missed.
The global diabetes epidemic has created unprecedented demand for better blood glucose tracking solutions, with over 422 million people worldwide living with diabetes according to World Health Organization statistics. Understanding the fundamentals of blood sugar monitoring remains essential for everyone living with diabetes regardless of which technology they choose. The monitoring protocols recommended by public health agencies emphasize regular glucose checks tailored to individual treatment regimens and diabetes types. Learning proper monitoring techniques, understanding target ranges, and knowing when to seek medical attention for abnormal readings forms the foundation of successful diabetes management. Educational resources provided by health authorities help people navigate the complexities of glucose monitoring while making informed decisions about incorporating continuous monitoring technologies into their care routines. In the United States alone, approximately 38 million adults have diabetes, and another 98 million have prediabetes, creating a massive market for innovative monitoring technologies. Traditional finger-stick blood glucose meters have served as the standard for decades, but their inherent limitations drive both patients and researchers toward revolutionary alternatives. The pain and inconvenience of frequent finger pricks leads to poor adherence, with studies showing that many people who check their glucose regularly take no action when results indicate high or low levels, rendering the discomfort essentially pointless. More critically, finger-stick tests provide only momentary snapshots of glucose levels, missing the dangerous fluctuations that occur between measurements and leaving people vulnerable to life-threatening hypoglycemic episodes during sleep or other periods without testing.
Before delving into the article, watch this video which highlights the truth about the accuracy of smartwatches in measuring blood sugar without finger pricks:
The year 2026 represents a pivotal moment in diabetes technology, with three distinct approaches competing for dominance in the glucose monitoring landscape. Smartwatch manufacturers promise the ultimate convenience of wrist-worn devices that could potentially measure glucose alongside heart rate and activity tracking, though regulatory approval remains elusive and technical challenges persist. Patch-based continuous glucose monitors have achieved mainstream acceptance with FDA clearance, offering proven accuracy and real-time alerts that have revolutionized diabetes management for hundreds of thousands of users. Implantable sensors push the boundaries further with year-long monitoring capabilities that eliminate frequent sensor changes, though surgical requirements and higher costs create barriers to widespread adoption. Each technology offers distinct advantages and limitations, and understanding these differences has become crucial for the millions of people seeking to escape the tyranny of constant finger-pricks.
The stakes extend far beyond mere convenience, as continuous glucose monitoring has demonstrated profound clinical benefits that directly impact health outcomes and quality of life. Research published by the Juvenile Diabetes Research Foundation showed that Type 1 diabetes patients using continuous glucose monitors achieved hemoglobin A1C reductions of 0.53 percent compared to those relying solely on traditional finger-stick monitoring, with even greater improvements among those who wore their sensors consistently. Patient advocacy organizations and diabetes education resources play crucial roles in translating complex medical research into practical guidance that helps people make informed technology decisions. Independent reviews of glucose monitoring devices provide valuable perspectives on real-world performance, user experiences, and practical considerations beyond clinical trial results. Educational content addressing common questions about device selection, troubleshooting, and optimization helps users get maximum benefit from their chosen monitoring technologies. These resources complement healthcare provider guidance by offering peer perspectives and detailed technical information that supports confident decision-making. Time spent in healthy glucose ranges increased dramatically, from baseline averages around 58 percent to over 70 percent for dedicated CGM users, translating directly into reduced risk of devastating long-term complications including diabetic retinopathy, nephropathy, and cardiovascular disease. The American Diabetes Association’s 2026 Standards of Care now explicitly recommend continuous glucose monitoring at diabetes onset and anytime thereafter for anyone who could benefit from its use, reflecting the compelling evidence base that has accumulated over recent years.
Professional medical organizations establish clinical practice guidelines based on comprehensive evidence reviews that inform healthcare provider recommendations and insurance coverage decisions. The annual updates to diabetes care standards reflect new research findings, emerging technologies, and evolving understanding of optimal management strategies. These evidence-based recommendations help standardize care quality across different healthcare settings while allowing flexibility for individual patient needs and circumstances. Staying current with the latest clinical guidelines ensures that both patients and providers understand current best practices for incorporating glucose monitoring and other diabetes technologies into comprehensive treatment plans.
The technological evolution has accelerated rapidly, with innovations emerging across all three monitoring categories. Smartwatch developers continue refining optical sensors, machine learning algorithms, and multimodal approaches that combine various physiological signals to estimate glucose levels, despite regulatory setbacks and accuracy concerns. Patch-based systems have shrunk to the size of stacked coins while extending wear times to fourteen days and reducing calibration requirements, with some models now available over-the-counter without prescription for wellness-focused users. Implantable sensors have overcome early biocompatibility challenges through advanced coating technologies that reduce foreign body responses, enabling the extended wear times that make them viable alternatives to frequent patch replacements. The convergence of these technologies with artificial intelligence, automated insulin delivery systems, and cloud-based data platforms is creating an ecosystem of connected diabetes management tools that would have seemed like science fiction just a decade ago.
Understanding the current state of each technology category requires examining not just their technical specifications but also their real-world performance, user experiences, regulatory status, and integration into comprehensive diabetes care strategies. The FDA has established increasingly sophisticated frameworks for evaluating glucose monitoring devices, moving beyond simple accuracy metrics toward holistic assessments of clinical outcomes, user burden, and integration capabilities. The European Union’s stricter Medical Device Regulation has created additional hurdles that companies must clear before bringing products to market, potentially delaying innovation but also ensuring safety and efficacy. Healthcare providers now navigate complex decision-making processes when recommending monitoring technologies, balancing individual patient needs, insurance coverage limitations, technical literacy, and personal preferences while staying current with rapidly evolving evidence and device capabilities.
The economic implications ripple throughout the healthcare system, as diabetes and its associated complications comprise roughly one-quarter of total healthcare spending in the United States. Implementing preventative monitoring strategies that catch glucose excursions before they cause damage could generate significant long-term cost savings while improving quality of life for millions. Insurance coverage policies have evolved to reflect the clinical evidence supporting continuous glucose monitoring, with Medicare, Medicaid, and most private insurers now covering devices for Type 1 diabetes and insulin-dependent Type 2 diabetes patients. However, the 2026 Medicare Home Health Rule introduced concerning changes that may limit access to specific devices, highlighting ongoing tensions between cost containment and optimal care delivery. For the growing number of people with prediabetes or non-insulin-dependent Type 2 diabetes who want glucose insights for lifestyle optimization, over-the-counter options are creating new markets outside traditional prescription channels.
Understanding Current Glucose Monitoring Technologies
The fundamental challenge underlying all glucose monitoring approaches stems from the biological complexity of actually measuring glucose concentrations in living tissue without disrupting that tissue. Traditional blood glucose meters solve this problem through the most direct method possible by obtaining an actual blood sample via finger-prick, then using enzymatic reactions to quantify glucose concentration with reasonable accuracy. The glucose oxidase or glucose dehydrogenase enzymes react specifically with glucose molecules, generating electrical signals proportional to glucose levels that can be converted into numerical readings. This approach works reliably because blood glucose concentrations directly reflect the systemic glucose levels that determine physiological responses, making measurements straightforward despite the invasive sample collection process. The National Glycohemoglobin Standardization Program ensures that devices meet traceability standards to the Diabetes Control and Complications Trial reference assay, providing consistency across different meter brands and models.
Continuous glucose monitors shift the measurement location from capillary blood to interstitial fluid, the liquid surrounding cells just beneath the skin surface, using tiny sensors inserted approximately five millimeters under the skin. These sensors employ similar enzymatic detection principles but must operate continuously in a hostile biological environment for days or weeks rather than processing a single controlled sample. The glucose oxidase coatings on the electrode surface react with glucose molecules diffusing from nearby capillaries, generating electrons that flow to the sensor electronics and get converted into glucose readings every one to fifteen minutes depending on the system. This represents a remarkable engineering achievement, but it introduces several complications including the five to ten minute lag time as glucose diffuses from blood vessels into interstitial fluid, potential interference from medications or other chemicals, and the complex foreign body responses that can degrade sensor performance over time.
The physiological relationship between blood glucose and interstitial fluid glucose remains relatively stable during steady-state conditions, allowing CGM systems to provide clinically useful information for most diabetes management decisions. However, during periods of rapid glucose change such as after meals or during hypoglycemia treatment, the lag time between blood and interstitial measurements can create discrepancies that require clinical awareness. When blood glucose rises quickly after eating, interstitial fluid levels may still reflect the lower pre-meal values, potentially causing users to dose excessive insulin based on outdated information. Conversely, during rapidly falling glucose levels, interstitial readings may overestimate actual blood glucose, leading to unnecessary carbohydrate consumption that drives glucose levels too high. Healthcare providers and experienced CGM users learn to anticipate these dynamics, using trend arrows and rate-of-change indicators to make informed decisions rather than relying solely on numerical values.
The sensor accuracy has improved dramatically across successive device generations, with modern systems achieving Mean Absolute Relative Difference values around nine percent compared to laboratory reference measurements. This metric reflects the average absolute difference between CGM readings and actual blood glucose levels, with lower numbers indicating better accuracy. For context, point-of-care finger-stick meters typically achieve MARD values between seven and twelve percent, meaning today’s best continuous glucose monitors match or exceed traditional monitoring accuracy while providing hundreds of readings daily instead of a handful. The FDA’s special controls for integrated continuous glucose monitors establish performance standards that devices must meet for automated insulin delivery integration, ensuring sufficient accuracy and reliability for making therapy decisions without confirmatory finger-sticks. These regulatory frameworks have evolved as the technology matured, balancing innovation with patient safety in ways that continue shaping development priorities.
Sensor calibration requirements have decreased or disappeared entirely in current generation devices, eliminating a major barrier to adoption and reducing user burden substantially. Earlier CGM systems required users to perform finger-stick calibrations twice daily to maintain accuracy, essentially combining the inconvenience of traditional monitoring with the discomfort of wearing a sensor. Modern factory-calibrated sensors use improved manufacturing processes and algorithmic corrections to deliver reliable readings without user calibration, though some situations still warrant confirmatory finger-sticks when readings seem inconsistent with symptoms or during medication changes. The calibration-free approach dramatically improved adoption rates, particularly among people who had avoided CGM specifically because of the perceived dual burden of finger-sticks plus sensor wear.
Shop on AliExpress via link: wholesale-sensor-adhesive-patches
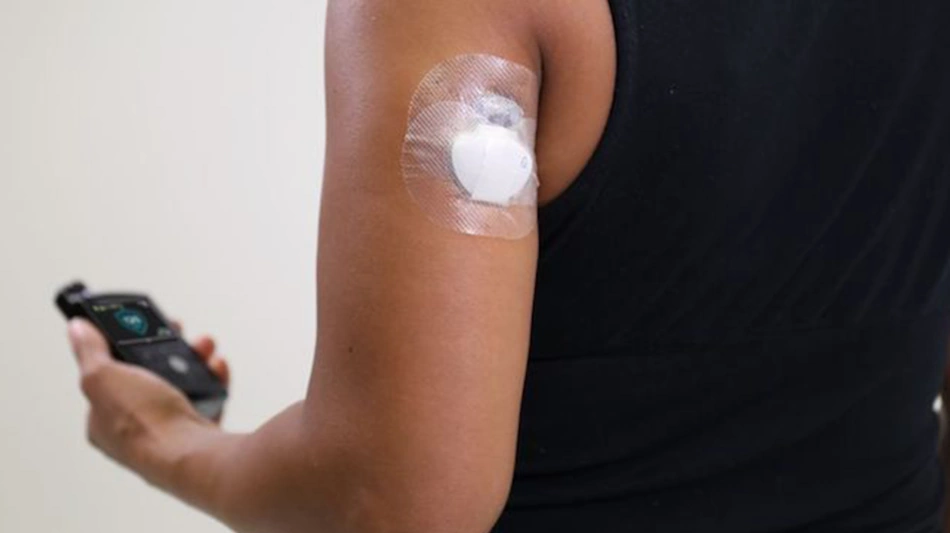
The sensor insertion process varies across device platforms but generally involves spring-loaded applicators that automatically insert the thin sensor filament beneath the skin with a quick needle stick. Users typically report minimal discomfort during insertion, far less than the cumulative pain of multiple daily finger-pricks, though psychological barriers around deliberately inserting a device into one’s body can create initial resistance. The insertion sites rotate between approved body locations, typically the back of the upper arm or abdomen, with sensors secured by adhesive patches designed to withstand showering, swimming, and vigorous physical activity. Adhesive-related skin irritation affects some users, creating redness or itching that can be managed with barrier wipes, alternative tape formulations, or strategic site rotation to allow healing between sensor applications.
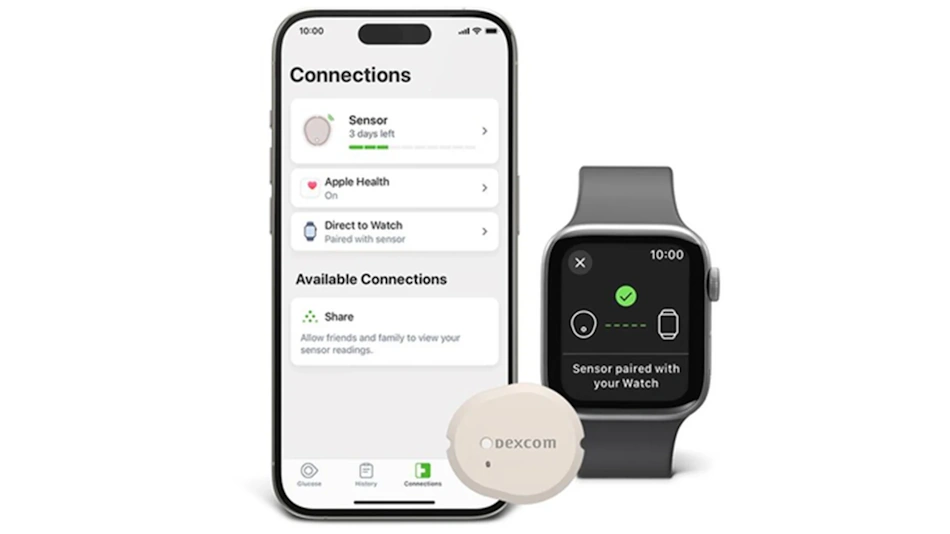
Transmitter technologies have evolved from bulky proprietary receivers to smartphone-based systems that leverage existing consumer devices, dramatically improving user experience and data accessibility. The evolution of continuous glucose monitoring systems has transformed diabetes care through technological innovations that make devices smaller, more accurate, and easier to use. Research supported by federal health institutes continues advancing sensor technologies, improving biocompatibility, and developing novel approaches to glucose measurement. Understanding how these systems work helps patients and healthcare providers make informed choices about incorporating continuous monitoring into comprehensive diabetes management strategies. The scientific foundation underlying modern glucose sensors combines electrochemistry, materials science, and sophisticated algorithms to deliver reliable measurements in challenging biological environments. Modern CGM systems transmit readings via Bluetooth Low Energy to compatible smartphones, smartwatches, or dedicated receivers, with data automatically uploaded to cloud platforms for healthcare provider review and retrospective analysis. The broader landscape of health monitoring wearables continues evolving beyond glucose-specific devices to encompass comprehensive fitness and wellness tracking platforms. Understanding the full range of available health monitoring technologies helps users make informed decisions about which devices best meet their individual needs and integrate effectively with existing diabetes management tools. Modern wearable technology ecosystems often allow multiple devices to work together, sharing data and providing holistic health insights that inform better lifestyle choices. Evaluating different fitness trackers and health monitoring options alongside glucose monitoring systems enables creation of personalized technology stacks optimized for individual health goals and management priorities.This connectivity enables family members to remotely monitor glucose levels for children or elderly relatives, providing peace of mind and enabling rapid intervention during emergencies. The integration with insulin pumps creates the foundation for automated insulin delivery systems that use CGM data to modulate insulin infusion rates, moving closer to artificial pancreas functionality that could transform diabetes management from constant vigilance to automated background process.
Alert systems represent another crucial advancement, with customizable thresholds for high and low glucose warnings that can prevent dangerous excursions before they cause serious problems. Users set alert levels based on their individual management goals and hypoglycemia awareness, receiving vibration, audio, or visual notifications when glucose trends outside target ranges or changes rapidly. The urgent low alert set at 55 milligrams per deciliter provides critical protection against severe hypoglycemia, while trend arrows indicating rapid glucose changes help users anticipate and prevent threshold violations before they occur. Some systems include predictive alerts that warn users twenty to thirty minutes before projected threshold crossings, enabling proactive intervention rather than reactive response to already-developed problems.
Smart Watch Integration: The Reality vs The Hype
The consumer technology industry’s fascination with non-invasive glucose monitoring has generated enormous hype and corresponding disappointment as reality consistently falls short of ambitious promises. Apple, Samsung, Huawei, and numerous startups have invested billions of dollars in developing optical, electromagnetic, and other sensing technologies that could theoretically measure glucose through intact skin without requiring sensor insertion. The potential market value drives continued investment despite repeated failures, as successfully cracking the non-invasive monitoring challenge would instantly obsolete current CGM technologies and capture massive market share. However, the fundamental physics and biology create barriers that prove far more difficult than early optimism suggested, leading to a graveyard of failed companies and abandoned projects that attempted various approaches without achieving regulatory approval or demonstrated clinical utility.
The FDA issued a stark safety communication in February 2024 explicitly warning consumers about smartwatches and smart rings claiming to measure blood glucose levels without piercing the skin. The regulatory landscape for glucose monitoring technologies continues evolving as manufacturers push boundaries with innovative sensing approaches. Medical device clearance processes established by the FDA ensure that approved systems meet rigorous safety and efficacy standards before reaching consumers. These regulatory frameworks balance encouraging innovation with protecting patient safety, requiring extensive clinical testing demonstrating accuracy across diverse populations and conditions. Healthcare providers and patients benefit from understanding the distinction between FDA-cleared devices that have undergone regulatory review and unapproved products making unsubstantiated claims about glucose measurement capabilities. The agency emphasized that no such devices had received FDA authorization, clearance, or approval, and that using these unvalidated products could lead to dangerously inaccurate measurements resulting in wrong treatment decisions. The warning specifically distinguished between unauthorized devices claiming to independently measure glucose and legitimate smartwatches that display data from FDA-approved continuous glucose monitors via wireless connection. For people with diabetes who rely on accurate glucose information to make insulin dosing decisions, using an unapproved smartwatch that provides false readings could lead to severe hypoglycemia from insulin overdoses or dangerous hyperglycemia from inadequate treatment, both potentially life-threatening situations.
The technical challenges underlying truly non-invasive optical glucose monitoring stem from the extremely weak glucose absorption signals that must be detected through multiple layers of skin tissue with varying properties. Glucose molecules represent less than one percent of skin composition, generating absorption patterns that easily get overwhelmed by interference from water, proteins, lipids, melanin, and other compounds present in far higher concentrations. Skin thickness, hydration status, temperature, and individual variations in tissue composition all affect optical measurements in ways that create unacceptable accuracy variability across different people and conditions. Early research used near-infrared spectroscopy, Raman spectroscopy, optical coherence tomography, and other optical techniques to detect glucose-related signals, but translating laboratory proof-of-concept demonstrations into reliable clinical devices proved vastly more difficult than anticipated.
Machine learning algorithms represent the latest attempt to overcome these fundamental physics limitations by combining multiple sensor inputs and learning complex patterns that correlate with actual glucose levels. The technical and engineering communities continue advancing biosensor technologies through fundamental research published in peer-reviewed journals and presented at scientific conferences. Innovations in wearable sensors, signal processing algorithms, and biocompatible materials emerge from multidisciplinary collaboration between electrical engineers, biomedical researchers, and clinical investigators. Understanding the underlying engineering principles helps appreciate both the remarkable capabilities of current devices and the substantial challenges facing next-generation development efforts. The intersection of biomedical engineering with glucose monitoring creates exciting opportunities for breakthrough technologies that could transform diabetes care while demanding rigorous validation before clinical deployment. These approaches measure heart rate variability, skin temperature, bioimpedance, activity levels, and other physiological parameters that change in response to glucose fluctuations, then use sophisticated algorithms to estimate glucose concentrations from these indirect signals. Some research demonstrations have shown promising correlation under controlled conditions, but real-world performance degrades significantly when faced with the tremendous variability in human physiology, behaviors, and environmental factors. A machine learning model trained on data from healthy young adults may completely fail when applied to older individuals with different metabolic characteristics, and models trained on one individual often don’t transfer well to others.
Samsung announced in January 2024 their intention to add non-invasive glucose monitoring to Galaxy smartwatches and smart rings, describing it as a significant investment priority alongside continuous blood pressure monitoring. The company’s digital health division suggested they were making substantial progress toward bringing these capabilities to market, generating considerable media attention and consumer excitement. However, as of 2026, no Samsung devices with FDA-cleared non-invasive glucose monitoring have reached consumers, illustrating the persistent gap between ambitious announcements and delivered products. The regulatory pathway requires demonstrating accuracy comparable to approved glucose monitoring devices across diverse populations and conditions, a standard that no optical technology has yet achieved in peer-reviewed clinical trials.
Apple’s secret glucose monitoring project has been the subject of intense speculation and reporting for over a decade, with various leaks suggesting they’re developing optical sensors for integration into Apple Watch. The company has reportedly assembled teams of biomedical engineers and acquired multiple startups working on non-invasive sensing technologies, investing enormous resources into solving the technical challenges. However, Apple’s legendary secrecy makes it impossible to verify actual progress, and no credible evidence suggests they’re closer to regulatory approval than competitors who have been more transparent about their development efforts. Industry analysts increasingly skeptical that even Apple’s massive resources and engineering talent can overcome the fundamental physical limitations that have defeated dozens of other companies attempting similar approaches.
The distinction between displaying CGM data and actually measuring glucose independently has become critically important as legitimate integration options proliferate. Current Dexcom G7, FreeStyle Libre, and other FDA-approved CGM systems can transmit readings to compatible smartwatches including Apple Watch, Garmin devices, and select Android Wear models, allowing users to view their glucose levels and trends without pulling out their phones. This integration provides genuine value through improved convenience and awareness, particularly during activities where phone access is impractical. Users receive alerts directly on their wrist, can glance at current readings and trend arrows discreetly throughout the day, and benefit from all the accuracy and reliability of clinically validated CGM sensors while enjoying smartwatch convenience. However, these capabilities depend entirely on the separate FDA-approved CGM sensor doing the actual glucose measurement work.
Research into noninvasive hypoglycemia detection represents a more modest but potentially achievable goal compared to continuous glucose measurement. Studies published in Diabetes Care demonstrated that machine learning algorithms analyzing smartwatch data from heart rate, heart rate variability, and accelerometer sensors could detect hypoglycemic episodes with approximately 85 percent sensitivity in people with diabetes. Emerging research into wearable health technologies explores novel sensing modalities and computational approaches that could complement or enhance current glucose monitoring methods. Scientific investigations documented in medical and engineering journals examine the correlation between various physiological parameters and glucose fluctuations, seeking indirect measurement approaches that might avoid the limitations of direct glucose sensing. While many of these experimental technologies remain in research phases far from commercial availability, they demonstrate the ongoing innovation addressing unmet needs in diabetes monitoring. Critical evaluation of early-stage research helps maintain realistic expectations while appreciating the scientific progress toward improved monitoring solutions. The physiological changes during hypoglycemia including increased heart rate, altered heart rate variability patterns, and sometimes tremors or unusual movement create detectable signatures that don’t require measuring glucose directly. While this approach couldn’t replace continuous glucose monitoring for comprehensive diabetes management, it could provide valuable safety alerts warning users to check their glucose when hypoglycemia might be developing, particularly useful for people with impaired hypoglycemia awareness who don’t experience typical warning symptoms.
Patch-Based CGM Systems: The Current Gold Standard
The FreeStyle Libre system revolutionized diabetes monitoring by introducing factory-calibrated sensors that eliminated finger-stick calibrations, removing a major barrier that had prevented widespread CGM adoption. When Abbott received FDA approval for the original FreeStyle Libre in 2017, it represented the first continuous glucose monitoring system available in the United States that required no calibration finger-sticks, making the technology accessible to millions of people who found traditional CGM too burdensome. The system uses a sensor applied to the back of the upper arm that reads glucose levels through a quick scan with a handheld reader or smartphone, providing eight hours of glucose history and trend information with each scan. This intermittently scanned approach initially required users to actively scan rather than receiving automatic continuous updates, but subsequent generations evolved toward real-time alerting capabilities that matched traditional CGM functionality.
The FreeStyle Libre 3 Plus represents Abbott’s latest advancement, featuring the smallest and thinnest CGM sensor available as of 2026 at roughly the size of two stacked pennies. The sensor provides glucose readings every minute, significantly more frequent than previous models, transmitted automatically to users’ smartphones without requiring manual scanning. The 14-day wear time matches earlier versions, but accuracy improvements and miniaturization make the device less obtrusive and more comfortable for extended wear. An urgent low glucose alarm automatically activates at 55 milligrams per deciliter, providing critical protection against severe hypoglycemia even for users who haven’t set up custom alerts. The FDA approved Libre 3 Plus for use in people with diabetes ages two and above, expanding access to pediatric populations where early adoption of continuous monitoring can establish healthier management patterns and prevent long-term complications.
Dexcom has established itself as the innovation leader in CGM technology through successive generations that pushed accuracy, wear time, and integration capabilities. The Dexcom G7 introduced in recent years offers a completely integrated sensor-transmitter design that simplified application and reduced profile compared to earlier models with separate removable transmitters. The twelve-hour warm-up period that plagued earlier Dexcom systems dropped to just thirty minutes in G7, meaning users could switch sensors and return to receiving readings with minimal interruption. Accuracy reached new heights with MARD values under eight percent, surpassing many finger-stick meters and providing confidence for making all diabetes management decisions without confirmatory testing. The ten-day wear time balances sensor performance with practical replacement frequency, though some users report successful extended wear beyond the approved duration.
Integration with automated insulin delivery systems represents perhaps the most significant advantage of current generation patch-based CGMs, enabling closed-loop diabetes management that was impossible with finger-stick monitoring alone. The FDA created specific regulatory pathways for integrated CGM systems that meet stringent performance criteria for automated insulin delivery integration, establishing the foundation for hybrid artificial pancreas devices that automatically adjust insulin delivery based on real-time glucose data. Dexcom G6 became the first FDA-cleared integrated CGM, paving the way for partnerships with insulin pump manufacturers including Tandem, Medtronic, and Insulet. These systems use sophisticated control algorithms to increase insulin delivery when glucose trends high and reduce or suspend delivery when glucose drops, dramatically reducing the cognitive burden of constant diabetes management while improving time in target range and reducing hypoglycemia.
The American Diabetes Association’s 2026 Standards of Care marked a watershed moment by recommending automated insulin delivery systems as the preferred insulin delivery method for people with Type 1 diabetes and many Type 2 diabetes patients. This strong recommendation reflects accumulating evidence that AID systems consistently outperform even highly motivated individuals using multiple daily injections with separate CGM, particularly during overnight hours when manual management requires sleep interruption. The standards removed previous barriers including requirements for C-peptide testing or documented duration of diabetes before accessing technology, recognizing that early adoption provides maximum benefit. Insurance coverage has expanded correspondingly, though the 2026 Medicare Home Health Rule introduced concerning changes that may restrict which specific devices Medicare beneficiaries can access.
The over-the-counter CGM market emerged in 2024 when the FDA cleared Dexcom Stelo as the first continuous glucose monitor available without prescription. Stelo targets people 18 and older with Type 2 diabetes not using insulin or those interested in glucose insights for general wellness purposes, opening continuous monitoring to populations previously excluded from prescription CGM access. The system uses the same basic sensing technology as prescription Dexcom devices but with modified software and packaging for direct-to-consumer sales. This regulatory innovation recognizes that glucose monitoring benefits extend beyond insulin dosing decisions to include dietary insights, exercise planning, and metabolic health optimization for people without diabetes. Companies including Levels, Veri, and Supersapiens have built businesses around wellness-focused glucose monitoring, promising better health, nutrition optimization, and performance enhancement based on continuous glucose data.
Sensor accuracy requires consideration of multiple factors beyond simple MARD statistics, as real-world performance depends on proper application technique, site selection, and individual physiological responses. The initial 24-48 hours after sensor insertion often show reduced accuracy as the foreign body response generates inflammation and tissue changes around the sensor membrane, though improved biocompatibility coatings have minimized this effect in current generation devices. Compression of the sensor site during sleep can temporarily affect readings, particularly for sensors placed on body locations prone to pressure during typical sleeping positions. Dehydration, certain medications including acetaminophen in older sensor models, and rapidly changing glucose levels can all contribute to reading discrepancies that warrant confirmatory finger-stick testing before making critical treatment decisions.
The adhesive patch technology securing sensors to skin has evolved substantially as manufacturers addressed user complaints about skin irritation, sensor loss during activities, and challenges removing stubborn adhesive residue. Modern patches balance strong adhesion for reliable wear with skin-friendly formulations that minimize irritation, though individual sensitivities vary widely and some users require barrier wipes or alternative adhesive strategies. Water resistance ratings enable swimming and showering throughout the approved wear period, with most sensors rated for submersion up to three feet for thirty minutes. Athletes report successful use during vigorous exercise, though some apply additional overlay patches to prevent sensor dislodgement during contact sports or activities involving significant sweating and friction.
Cost comparisons between different patch-based systems require examining total system costs including sensors, transmitters, receivers or compatible smartphones, and subscription fees for cloud data platforms. The FreeStyle Libre systems generally cost less than Dexcom equivalents when comparing out-of-pocket expenses, though insurance coverage substantially reduces costs for prescription users meeting medical necessity criteria. Over-the-counter CGMs like Stelo follow different pricing models aimed at broader consumer markets, with monthly subscriptions typically ranging from $89 to $99 for the sensors needed. These prices remain prohibitive for many people who might benefit from continuous monitoring but don’t have insurance coverage or medical necessity for prescription options, highlighting ongoing access challenges despite technological progress.
Implantable Sensors: Long-Term Solutions
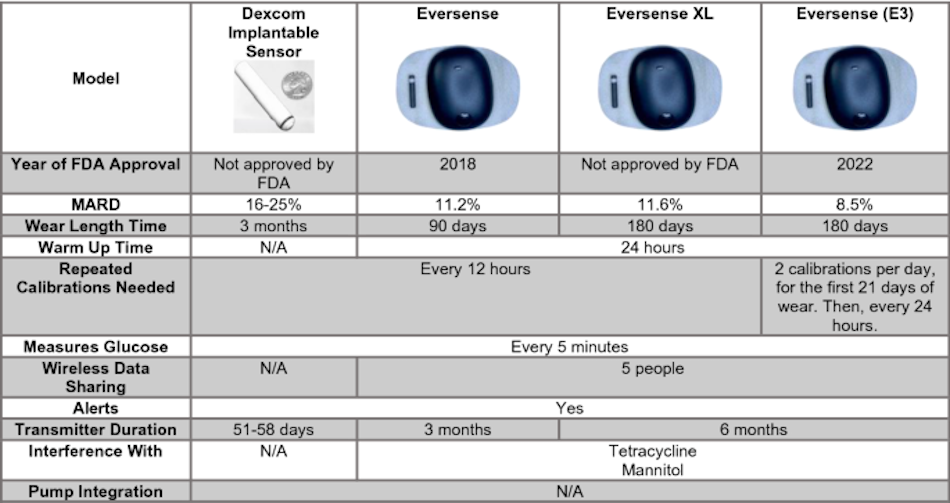
The Eversense CGM system from Senseonics introduced the first and currently only FDA-approved fully implantable continuous glucose monitoring sensor designed for long-term wear. Unlike patch-based systems where users replace disposable sensors every 7-14 days, Eversense uses a small fluorescence-based sensor implanted beneath the skin of the upper arm during a brief in-office procedure performed by healthcare professionals. The sensor itself remains implanted for up to 365 days in the latest Eversense 365 model, while a removable smart transmitter adheres externally over the implant site and communicates wirelessly with the user’s smartphone. This design enables users to temporarily remove the transmitter for contact sports, showering, or other activities where external devices might be problematic, then reattach it to resume glucose monitoring without interrupting the internal sensor function.
The implantation procedure takes approximately five minutes and involves local anesthesia at the insertion site, followed by a small incision through which the sensor is placed in the subcutaneous tissue. Healthcare providers typically use ultrasound guidance to ensure proper sensor positioning and depth, then close the incision with sterile strips or sutures that dissolve over several days. Patients experience mild discomfort and temporary soreness similar to a minor cut or injection, but the procedure avoids general anesthesia or significant surgical intervention. After a 24-hour warm-up period, the sensor begins providing glucose readings every five minutes transmitted to the user’s smartphone via the external transmitter. At the end of the approved wear period, a similar brief procedure removes the expired sensor, and a new sensor can be immediately implanted in a different location or the same site after appropriate healing.
The fluorescence-based sensing mechanism used by Eversense differs fundamentally from the enzymatic electrochemical approach used by patch-based CGMs. The implanted sensor contains a glucose-responsive polymer embedded with fluorescent molecules that emit light when excited by LED pulses from the external transmitter. Glucose molecules penetrate the polymer and bind reversibly to the sensing chemistry, causing changes in fluorescence intensity that correlate with glucose concentrations. The transmitter measures these fluorescence changes and converts them into glucose readings using calibration algorithms. This optical approach offers potential advantages including reduced susceptibility to electrochemical interference and potentially longer sensor lifespan compared to enzymatic sensors that can degrade as enzyme activity diminishes over time.
Clinical studies supporting FDA approval demonstrated accuracy comparable to leading patch-based CGM systems with MARD values around nine percent across the full glucose range. One particularly notable finding showed significantly reduced false low alerts during nighttime hours compared to patch systems, an important advantage since excessive false alerts drive users to disable protective alarms entirely. The on-body vibration alerts built into the transmitter provide notifications for high and low glucose without requiring phone proximity, useful for people who may not always have their smartphone nearby or who sleep too deeply to hear phone alerts. The transmitter battery lasts approximately one day between charges, requiring nightly charging similar to smartphone routines that most users find manageable.
The extended wear time represents Eversense’s primary value proposition, eliminating 26-52 sensor replacement procedures annually compared to weekly or biweekly patch changes. For users who experience skin irritation from adhesives, anxiety about sensor insertion, or frustration with the ongoing hassle of frequent replacements, the year-long implant offers substantial quality of life improvements despite the surgical requirements. The total annual cost may actually decrease compared to frequent patch replacements when accounting for reduced sensor expenses, though individual cost comparisons depend heavily on insurance coverage specifics. Medicare and many private insurers cover Eversense for qualified patients, though coverage criteria and prior authorization requirements vary by plan.
Foreign body responses to implanted medical devices pose significant challenges that have limited the adoption of implantable glucose sensors. When any foreign object sits beneath the skin for extended periods, the immune system generates inflammation and eventually forms a fibrous capsule around the implant in an attempt to wall it off from surrounding tissue. This fibrosis can impair glucose diffusion to the sensor, degrading accuracy and potentially causing sensor failure before the approved wear period ends. Eversense addresses this challenge through specialized biocompatible coatings and polymer formulations designed to minimize inflammatory responses and maintain glucose permeability throughout the year-long implantation. Research into next-generation implants explores advanced coating materials including zwitterionic polymers that create hydration layers preventing protein adhesion and reducing cellular responses.
Infection risk accompanies any procedure that breaks the skin barrier, requiring careful sterile technique during both insertion and removal procedures. Healthcare providers follow strict protocols including skin preparation, sterile draping, and proper wound care instructions to minimize contamination. Patients receive guidance on monitoring for signs of infection including increasing pain, redness, warmth, or discharge from the incision site, with instructions to contact their healthcare team immediately if any concerning symptoms develop. Actual infection rates remain extremely low when procedures follow proper protocols, but the theoretical risk exceeds that of patch-based systems where users perform sensor insertion themselves without breaking the skin beyond the depth reached by the automated applicator needle.
The psychological and lifestyle factors influencing implant acceptance vary dramatically across individuals, with some people strongly preferring the implant approach while others find the surgical requirement unacceptable. People who have already undergone multiple surgical procedures for other medical conditions may view a simple sensor implantation as trivial, while those with surgical anxiety or needle phobias may find even minor procedures psychologically overwhelming. The ability to remove the transmitter for specific activities appeals to athletes, people who engage in contact sports, or those who simply dislike the visible external hardware of patch-based systems. However, the requirement to reattach the transmitter within certain time windows to avoid data gaps creates compliance demands that some users find burdensome compared to the set-and-forget nature of patch systems.
Insurance coverage and reimbursement policies for implantable CGMs lag behind patch-based systems in some markets, reflecting both the relative novelty of implants and potential concerns about cost-effectiveness given the higher upfront procedure costs. Prior authorization requirements may be more stringent for implants, requiring documentation of failed patch-based CGM trials or specific medical justifications for choosing implantable technology. Medicare policies have evolved to include Eversense coverage for beneficiaries meeting criteria for continuous glucose monitoring, though the 2026 Medicare Home Health Rule changes created uncertainty about future access. International markets show variable coverage, with some European countries providing robust implant access while others maintain restrictive policies favoring lower-cost patch options.
The development pipeline for next-generation implantable sensors includes systems targeting even longer wear times extending to multiple years, smaller form factors that minimize the implantation procedure invasiveness, and integration with insulin delivery systems for fully implanted closed-loop artificial pancreas devices. Researchers envision future implants that could monitor multiple biomarkers simultaneously including glucose, lactate, ketones, and other metabolites relevant to diabetes and general health management. The technical challenges remain substantial, particularly around maintaining sensor accuracy and stability over years rather than months, but successful solutions could eliminate external diabetes devices entirely and enable truly invisible disease management.
Accuracy Comparison: Which Technology Performs Best
Evaluating glucose monitoring accuracy requires understanding multiple metrics beyond simple correlation with laboratory reference measurements, as clinical utility depends on how discrepancies between measured and actual glucose levels affect treatment decisions and health outcomes. The Mean Absolute Relative Difference represents the most commonly reported accuracy metric, calculating the average percentage deviation between CGM readings and reference blood glucose measurements across all glucose ranges. Modern patch-based CGMs achieve MARD values between 8-10 percent, with the best performing systems like Dexcom G7 reaching under eight percent in clinical trials. For context, even laboratory reference methods show some variability, and home blood glucose meters typically achieve MARD values between seven and twelve percent, meaning current CGMs match or exceed the accuracy of traditional finger-stick monitoring that has served as the diabetes management standard for decades.
The Clarke Error Grid provides another critical assessment by categorizing glucose monitoring errors based on their clinical consequences rather than just numerical accuracy. Zone A contains readings within 20 percent of reference values or clinically acceptable ranges that would lead to appropriate treatment decisions. Zone B includes readings outside acceptable accuracy ranges but unlikely to cause harmful treatment errors. Zones C, D, and E represent progressively more dangerous errors that could lead to inappropriate treatment and potential harm. High-quality CGM systems place over 99 percent of readings in Clarke Error Grid zones A and B, with fewer than one percent falling into potentially problematic zones C through E. This clinical accuracy assessment often matters more than raw MARD statistics, as a system with slightly higher average error but excellent error distribution may actually perform better for real-world diabetes management.
Hypoglycemia detection represents perhaps the most critical safety consideration, as failure to alert users about dangerously low glucose levels can result in severe hypoglycemic episodes causing seizures, loss of consciousness, or even death in extreme cases. Current generation CGMs demonstrate excellent sensitivity for detecting glucose levels below 70 milligrams per deciliter, the threshold defining hypoglycemia for most clinical purposes. However, the interstitial fluid measurement lag during rapidly falling glucose can sometimes cause delayed alerts, with sensors showing higher readings than actual blood glucose during acute drops. The trend arrows and predictive algorithms partially compensate by alerting users to rapid decreases before threshold violations occur, but the inherent lag time creates theoretical windows where severe hypoglycemia could develop despite sensor readings remaining above alert thresholds. Healthcare providers counsel patients about this limitation and recommend treating hypoglycemia symptoms even when sensor readings seem adequate.
Hyperglycemia detection faces fewer safety challenges since high glucose develops more gradually and rarely causes immediate life-threatening situations, though chronic hyperglycemia drives long-term complications. CGM accuracy remains good throughout the hyperglycemic range, though percentage errors increase at very high glucose levels above 300-400 milligrams per deciliter. Some CGM systems truncate displayed readings at 400 or 500 milligrams per deciliter to avoid displaying measurements in ranges where accuracy becomes questionable, instead showing “HIGH” with instructions to confirm with finger-stick testing. This conservative approach prevents users from making insulin dosing decisions based on potentially inaccurate extreme high readings while still alerting them to the serious hyperglycemia requiring immediate intervention.
Individual variability in CGM accuracy complicates comparisons and contributes to different user experiences with identical technology. Factors including body composition, skin thickness, hydration status, and individual immune responses to the foreign sensor can all affect performance in ways that vary between people and even within the same person over time. Some users report excellent accuracy with particular sensor brands while others experience frequent discrepancies requiring confirmatory finger-sticks. Age-related changes in skin properties may affect sensor performance differently in pediatric versus adult versus elderly populations, though all approved systems demonstrate acceptable accuracy across their labeled age ranges.
The first 24 hours after sensor insertion typically show reduced accuracy as the initial inflammatory response to sensor placement affects the tissue environment around the sensing membrane. Users often notice readings that seem higher or lower than finger-stick confirmations during this warm-up period, though the discrepancies usually resolve as inflammation subsides and tissue equilibrates. Some healthcare providers recommend avoiding major treatment decisions based solely on CGM readings during the first day after sensor application, instead using finger-stick confirmations for insulin dosing until sensor performance stabilizes. Manufacturers continue researching biocompatible coatings and sensor designs that minimize this break-in period, with some newer systems showing improved early accuracy compared to earlier generations.
Calibration requirements affect both user burden and accuracy potential, with factory-calibrated systems trading some theoretical accuracy for dramatically improved convenience. Earlier CGM systems required twice-daily calibration finger-sticks to maintain accuracy, essentially demanding users perform traditional glucose monitoring while also wearing sensors. The calibration process adjusted sensor algorithm parameters based on confirmed blood glucose values, theoretically improving accuracy by accounting for individual variation. However, user calibration also introduced opportunities for error if finger-stick values were inaccurate or if users performed calibrations during periods of rapidly changing glucose when blood and interstitial measurements diverged. Factory calibration eliminates these user error sources and removes a major adoption barrier, though some users of older systems reported achieving slightly better accuracy with frequent calibrations.
The regulatory requirements for CGM accuracy have evolved as technology improved and clinical experience accumulated. The FDA established integrated CGM special controls defining performance criteria that devices must meet for use in automated insulin delivery systems without confirmatory finger-sticks. These stringent requirements ensure that CGMs used for automated therapy decisions maintain sufficient accuracy and reliability across diverse populations and conditions. The standards include specific MARD thresholds, performance in hypoglycemic and hyperglycemic ranges, and stability over the approved wear period. Devices meeting these criteria can be labeled as integrated CGMs and used in closed-loop systems, while those falling short may still receive approval for adjunctive use requiring finger-stick confirmations before treatment decisions.
Post-market surveillance and real-world evidence studies increasingly inform understanding of CGM accuracy beyond controlled clinical trials. Healthcare providers report that actual field performance sometimes differs from trial results, with some users experiencing better accuracy than expected while others encounter challenges not evident in research settings. The FDA encourages reporting of device malfunctions, inaccurate readings, and other performance issues through MedWatch, the agency’s adverse event reporting system. Manufacturers analyze these reports along with user data uploaded to cloud platforms to identify potential issues requiring software updates, manufacturing process changes, or in extreme cases product recalls to protect patient safety.
Cost Analysis and Insurance Coverage in 2026
The financial burden of diabetes management extends far beyond glucose monitoring devices to include insulin, other medications, medical appointments, complications treatment, and the indirect costs of lost productivity and reduced quality of life. Glucose monitors represent a relatively small but essential component of total diabetes expenses, with costs varying dramatically depending on technology choice, insurance coverage, and individual usage patterns. Traditional finger-stick monitoring using blood glucose meters and test strips typically costs between $400 and $1,200 annually for supplies at retail pricing, with exact figures depending on testing frequency and whether generic or brand-name products are chosen. Insurance coverage usually reduces out-of-pocket costs significantly, though copays and coinsurance percentages vary by plan specifics.
Continuous glucose monitoring systems carry substantially higher costs than traditional monitoring when comparing retail prices without insurance coverage. The sensor supplies alone for patch-based CGMs range from approximately $3,000 to $6,000 annually depending on the specific system and whether users replace sensors at exactly the approved wear time or achieve extended wear beyond labeled duration. Dexcom G7 sensors retail for around $230 for a pack of three 10-day sensors, translating to roughly $2,800 annually for sensor supplies. FreeStyle Libre systems generally cost less with sensor packs around $110 for two 14-day sensors, totaling approximately $1,900 yearly. These figures don’t include additional costs for compatible smartphones, smartwatches for data display, or potential subscription fees for advanced features in manufacturer apps.
Implantable CGM systems like Eversense follow different cost structures reflecting the year-long sensor lifespan and professional insertion requirements. The total cost for Eversense 365 including sensor, transmitter, insertion procedure, and removal procedure typically ranges from $3,500 to $5,000 annually depending on healthcare provider charges and geographic variation. While this may seem comparable to patch-based system costs, the convenience of just two procedures annually versus 26-52 sensor changes represents significant time savings and reduced hassle for users. Some individuals find the implant approach more cost-effective when accounting for the value of their time and the psychological benefit of not managing frequent sensor replacements.
Insurance coverage policies have evolved substantially as evidence supporting CGM clinical benefits has accumulated and diabetes technology organizations advocated for improved access. Most private insurance plans now cover continuous glucose monitors for Type 1 diabetes patients with minimal copays, recognizing these devices as medically necessary for safe diabetes management. Coverage criteria typically require documentation of diabetes diagnosis, current insulin use, and willingness to use the device regularly as demonstrated by trial periods or prescription history. Prior authorization processes vary by insurer but generally involve healthcare providers submitting medical necessity documentation explaining why CGM is appropriate for the specific patient.
Type 2 diabetes insurance coverage has expanded but remains more restricted than Type 1 coverage, usually requiring insulin use, frequent daily injections, or documented history of severe hypoglycemia. Some insurers cover CGM for Type 2 patients taking basal insulin who demonstrate engagement with diabetes self-management education and active use of glucose data to inform therapy adjustments. The 2026 ADA Standards of Care supporting broader CGM use including for patients on non-insulin therapies that can cause hypoglycemia may drive further coverage expansion, though insurance policy changes typically lag behind clinical guidelines by several years as payers evaluate cost-effectiveness and budget impact.
Medicare coverage for continuous glucose monitors has evolved significantly with multiple policy changes attempting to balance access with cost containment. Medicare Part B covers therapeutic CGM for beneficiaries meeting specific criteria including insulin therapy at least three times daily, four or more finger-sticks daily, and face-to-face visits with their treating physician every six months. The coverage criteria exclude many Type 2 diabetes patients who could potentially benefit from CGM but don’t meet the frequency of insulin administration requirements. The 2026 Medicare Home Health Rule introduced controversial changes to CGM and insulin pump coverage that raised concerns from the American Diabetes Association and advocacy groups about potential access restrictions for beneficiaries whose current devices may no longer qualify under new policies.
Medicaid coverage varies substantially by state as individual programs establish their own policies within federal guidelines. Some states provide generous CGM coverage comparable to Medicare or private insurance, while others maintain highly restrictive criteria or exclude coverage entirely. The state-by-state variability creates significant equity issues where identical patients living in different states face vastly different access to identical technology based solely on geography. Advocacy organizations continue pressing for standardized Medicaid coverage criteria that would ensure consistent access regardless of state residence.
Over-the-counter CGM pricing follows different models aimed at consumers paying entirely out-of-pocket without insurance coverage. Dexcom Stelo costs approximately $99 monthly for the sensors needed, marketing directly to wellness-focused consumers interested in glucose insights but not requiring prescription medical devices. This pricing deliberately undercuts prescription CGM out-of-pocket costs for uninsured individuals while remaining profitable for direct consumer sales. The subscription model provides predictable monthly expenses and includes smartphone app access, cloud data storage, and customer support without requiring healthcare provider involvement or insurance paperwork.
The total cost of ownership calculations must account for all system components beyond just sensors, including compatible devices, potential app subscription fees, and healthcare visit costs associated with CGM prescription and management. Users who already own compatible smartphones avoid separate receiver costs, while those requiring dedicated receivers add $350-400 to initial setup expenses. Some CGM manufacturers offer trade-in programs or discounts on receivers for users switching from competitor systems, reducing barriers to trying different technologies. The smartphone requirement creates digital divide concerns for populations without access to modern smartphones or data plans, potentially excluding lower-income individuals from CGM benefits despite meeting medical necessity criteria.
Financial assistance programs run by manufacturers, nonprofit organizations, and government programs help address access challenges for qualifying individuals. Dexcom and Abbott both operate patient assistance programs providing free or reduced-cost supplies to uninsured or underinsured people meeting income requirements. Diabetes advocacy organizations maintain directories of financial assistance resources and provide guidance navigating the complex landscape of coverage options, prior authorization processes, and appeals when insurance denies coverage. Healthcare providers increasingly employ dedicated diabetes educators or care coordinators who help patients navigate insurance coverage and identify assistance programs, recognizing that financial barriers significantly affect technology adoption and diabetes outcomes.
The Future: What’s Coming Next
The technological roadmap for glucose monitoring extends far beyond incremental improvements to current devices toward fundamentally new approaches that could finally achieve the elusive goal of truly non-invasive continuous monitoring. Research teams worldwide continue investigating optical spectroscopy methods despite decades of disappointments, with recent focus on mid-infrared wavelengths that show stronger glucose absorption signals than the near-infrared approaches that dominated earlier efforts. Proof-of-concept studies presented at the 2024 MIT Workshop on Noninvasive Glucose Monitoring demonstrated mid-infrared sensors achieving accuracy within 15 percent of reference measurements under controlled conditions, though substantial challenges remain before these laboratory results translate into wearable consumer devices suitable for regulatory approval.
Raman spectroscopy represents another optical approach gaining renewed attention as laser sources and detector technologies improve. The technique detects molecular vibrations specific to glucose molecules, potentially offering better specificity than absorption-based methods plagued by interference from other tissue components. Companies including Apollon are developing Raman-based continuous glucose monitoring devices leveraging signals from interstitial fluid beneath the skin surface, with clinical trials underway evaluating performance in people with diabetes. The challenge lies in detecting extremely weak Raman signals while minimizing laser power to safe levels that don’t cause tissue damage during continuous operation, requiring sophisticated signal processing and noise reduction algorithms.
Radio frequency sensing technologies employ electromagnetic waves to detect glucose-related changes in tissue dielectric properties, avoiding some limitations of optical approaches. The GlucoWise device under development uses 60 gigahertz radio waves applied to skin between the thumb and index finger or earlobe, measuring reflected signals that vary with tissue glucose concentrations. Early pilot studies with healthy volunteers showed promising correlation with reference glucose measurements, though testing in diverse diabetes populations under real-world conditions remains ongoing. The radio frequency approach faces challenges from interference by tissue hydration, temperature, and other factors that affect dielectric properties independently of glucose, requiring sophisticated algorithms to isolate glucose-specific signals from confounding variables.
Breath analysis represents a completely different approach exploiting the relationship between blood glucose and acetone concentrations in exhaled breath. During certain metabolic states including ketosis and diabetic ketoacidosis, the body produces ketones including acetone that appear in breath at levels correlating with metabolic status. Researchers have developed electronic nose devices containing sensor arrays that detect acetone and other volatile organic compounds in breath samples, using machine learning to predict blood glucose levels from the breath composition patterns. A 2022 IEEE Spectrum report described an e-nose achieving 90 percent accuracy in glucose level detection, though translating proof-of-concept to continuous monitoring devices suitable for diabetes management remains challenging.
Tear fluid glucose sensing attracted enormous attention when Google announced smart contact lens development in 2014, though the project was later discontinued after failing to achieve sufficient accuracy. Glucose concentrations in tear fluid correlate with blood glucose but at much lower levels and with complex lag times that vary depending on tear production rates and individual physiology. Despite Google’s withdrawal, researchers continue investigating tear glucose through alternative approaches including external collection devices and improved sensing chemistries. The non-invasive sample collection makes tear analysis attractive despite the technical challenges, and some companies remain actively developing tear-based monitoring systems targeting niche applications where perfect accuracy matters less than trend information.
Saliva and sweat-based glucose monitoring face similar challenges of extremely low glucose concentrations in these fluids combined with contamination from food, beverages, and individual variation in fluid production. Sweat-based sensors must contend with glucose levels one hundred times lower than blood, requiring extraordinarily sensitive detection systems prone to interference from sweat electrolytes, proteins, and metabolites. Some research groups have demonstrated glucose detection in sweat using advanced nanomaterial-based sensors, but achieving clinically useful accuracy for diabetes management remains elusive. The approaches may find application in wellness contexts where approximate glucose trends suffice rather than precise values needed for insulin dosing.
Implantable sensor improvements focus on extending functional lifespan beyond current year-long limits toward multi-year or even decade-long operation. The fundamental challenge involves maintaining sensor accuracy as biological responses including fibrosis and protein fouling progressively degrade glucose diffusion to sensing surfaces. Next-generation biocompatible coatings incorporating drug-eluting polymers could actively suppress inflammatory responses, while bio-inspired designs mimicking natural tissue structures might reduce foreign body recognition. Research into alternative sensing chemistries beyond traditional glucose oxidase enzymes explores more stable detection mechanisms less vulnerable to degradation over extended time periods.
Shop on AliExpress via link: wholesale-diabetes-monitoring-supplies
Artificial intelligence integration represents perhaps the most transformative near-term opportunity, with machine learning algorithms already enhancing CGM data interpretation and predictive capabilities. Current systems use relatively simple algorithms predicting glucose trends twenty to thirty minutes ahead based on recent patterns, but more sophisticated neural networks could potentially forecast glucose trajectories hours in advance by incorporating meal timing, activity, medication schedules, and physiological stress indicators. These predictions could enable more proactive diabetes management with interventions occurring well before problematic glucose excursions develop rather than reacting to already-present highs or lows.
The convergence of CGM with other wearable health sensors creates opportunities for holistic health monitoring platforms tracking multiple biomarkers simultaneously. The comparison between different health monitoring technologies reveals important considerations about data integration, user experience, and the relative merits of specialized versus general-purpose devices. Smart mirrors, fitness trackers, and dedicated medical devices each offer unique advantages in the evolving ecosystem of personal health technology. Understanding how these various technologies complement or compete with each other helps users build effective monitoring strategies that leverage the strengths of different approaches. The future of health monitoring likely involves sophisticated integration between multiple device types, creating comprehensive wellness platforms that provide insights impossible from any single technology alone. Future devices might combine continuous glucose monitoring with heart rate variability, blood pressure, hydration status, stress markers, and sleep quality metrics, using integrated data to provide comprehensive health insights and personalized recommendations. The correlation between various physiological parameters could improve glucose predictions, with cardiac data helping detect developing hypoglycemia, activity information refining mealtime dosing, and sleep patterns informing overnight basal adjustments.
Regulatory pathways for breakthrough technologies present significant challenges as traditional clinical trial designs may not adequately evaluate novel non-invasive approaches that work through indirect measurements. The FDA has shown willingness to create new device categories and approval pathways when warranted, as demonstrated by the over-the-counter CGM clearance for Dexcom Stelo. Future non-invasive devices may require innovative trial designs proving clinical utility even if they don’t achieve perfect correlation with laboratory reference measurements, particularly for wellness applications where approximate glucose awareness provides value without requiring precision needed for insulin dosing.
The economic disruption from successful truly non-invasive glucose monitoring could reshape the entire diabetes device industry virtually overnight, making current CGM systems obsolete and creating winner-take-all competition among technology developers. This enormous potential reward drives continued investment despite repeated failures, with optimists believing that breakthrough is always just around the corner while skeptics point to decades of unfulfilled promises. The reality likely lies somewhere between, with continued incremental progress making non-invasive monitoring increasingly viable for specific applications even if perfect systems for all diabetes management needs remain elusive.
Making Your Choice: Decision Framework
Selecting the optimal glucose monitoring approach requires careful consideration of individual medical needs, lifestyle factors, financial constraints, and personal preferences that vary dramatically between people even with identical diabetes diagnoses. Healthcare providers should engage patients in shared decision-making conversations exploring priorities and concerns rather than defaulting to one-size-fits-all prescriptions. Some people prioritize minimizing visible medical devices and might strongly prefer implantable sensors despite higher costs or surgical requirements, while others feel most comfortable with familiar patch-based systems they can control and replace themselves without healthcare provider involvement.
Medical necessity considerations start with diabetes type and treatment regimen, as insulin users face different monitoring needs than those managing through diet and oral medications alone. The 2026 ADA Standards recommend CGM for anyone who could benefit from continuous monitoring regardless of diabetes type or insulin status, dramatically expanding the population considered appropriate for this technology. Type 1 diabetes patients using insulin clearly benefit from CGM regardless of age or duration, with evidence supporting CGM initiation at diagnosis rather than waiting for problems to develop. Type 2 diabetes patients using intensive insulin therapy similarly demonstrate improved outcomes with CGM, while those on less intensive regimens may benefit from periodic CGM use for treatment optimization even without continuous wear.
Hypoglycemia awareness status critically affects monitoring recommendations, as people with impaired hypoglycemia awareness who don’t experience typical warning symptoms face substantially higher risk of severe episodes. These individuals benefit enormously from CGM alert systems that provide external warnings when their internal detection systems fail, making continuous monitoring potentially life-saving rather than merely convenient. Conversely, people with intact hypoglycemia awareness and well-controlled diabetes maintained with minimal insulin may not require continuous monitoring, instead using periodic CGM sessions for optimization or relying on strategic finger-stick testing.
Activity levels and lifestyle factors influence device selection among CGM options, with athletes and physically active individuals having different needs than sedentary users. Patch-based systems generally withstand vigorous exercise though some users report sensor loss during contact sports or activities involving significant impact or friction. Implantable systems offer advantages for water sports enthusiasts, swimmers, or people engaged in activities where external devices prove problematic, though the removable transmitter must be reattached within time windows to avoid data gaps. The aesthetic considerations around visible devices affect choices, with some people preferring small discrete sensors while others don’t care about visibility.
Technical literacy and comfort with smartphone technology create potential barriers, as all current CGMs require compatible smart devices and reasonable technological competence. Elderly individuals or those unfamiliar with smartphones may struggle with initial setup, data interpretation, and troubleshooting despite the diabetes management benefits. Healthcare teams can provide education and support addressing these barriers, and some users enlist family members to assist with technology management. The digital divide creates equity concerns where lower-income populations without smartphones or data plans cannot access devices increasingly considered standard diabetes care.
Insurance coverage specifics heavily influence practical decisions as out-of-pocket costs vary enormously depending on plan details. Patients should thoroughly investigate their specific coverage including deductibles, copays, coinsurance percentages, and preferred devices before making selections. Some insurers maintain preferred CGM brands with lower cost-sharing, steering patients toward specific manufacturers through financial incentives rather than clinical considerations. Prior authorization requirements and appeals processes add administrative burden that some people find overwhelming, though healthcare providers and diabetes educators increasingly help navigate these challenges.
Trial periods allow prospective CGM users to experience different systems before committing long-term, identifying issues or confirming benefits in real-world conditions rather than theoretical discussions. Many manufacturers and healthcare providers offer trial programs providing sensors at reduced cost or free for specified periods, allowing informed comparison shopping. The trial experience often reveals unexpected factors affecting satisfaction including adhesive tolerability, alarm preferences, smartphone app usability, and insertion process comfort that people couldn’t anticipate without hands-on experience.
The decision timeline doesn’t need to be permanent, as many users switch systems over time as their needs change, new technologies emerge, or insurance coverage shifts. Someone might start with patch-based CGM for initial experience then transition to implants once comfortable with continuous monitoring. Periodic reassessment ensures that monitoring approaches continue meeting evolving needs rather than persisting with outdated choices from inertia. Healthcare providers should revisit monitoring strategies regularly during routine visits, discussing whether current approaches remain optimal or whether changes might improve outcomes or quality of life.
Conclusion
The diabetes monitoring landscape of 2026 presents unprecedented options for people seeking accurate blood glucose tracking without the burden of constant finger-pricks that dominated diabetes management for decades. Patch-based continuous glucose monitors have matured into reliable, accurate systems that match or exceed traditional monitoring while providing hundreds of readings daily, trend information, and protective alerts that prevent dangerous glucose excursions. These devices have earned strong recommendations from the American Diabetes Association and achieved mainstream adoption among Type 1 diabetes patients, with expanding access for Type 2 diabetes and wellness populations. Implantable sensors offer compelling advantages for people prioritizing extended wear time and minimal external hardware, though surgical requirements and higher costs limit adoption compared to patch systems.
The promised future of truly non-invasive smartwatch glucose monitoring remains elusive despite enormous investment and repeated announcements from major technology companies. The FDA’s explicit warnings against unapproved devices claiming noninvasive measurement protect consumers from dangerous misinformation while highlighting the persistent gap between marketing hype and regulatory reality. Current smartwatch integration with approved CGM systems provides genuine value through improved data access and convenience, but these capabilities depend entirely on separate FDA-cleared sensors doing actual glucose measurement work. The fundamental physics challenges preventing accurate optical glucose measurement through intact skin show no signs of imminent breakthrough despite continued research efforts.
Making informed monitoring choices requires honest conversations between patients and healthcare providers exploring individual needs, preferences, constraints, and priorities rather than defaulting to prescriptive recommendations. The substantial clinical evidence supporting continuous glucose monitoring justifies strong consideration for anyone with diabetes who could benefit from improved glucose awareness, while financial and access barriers demand creative problem-solving to ensure equitable access regardless of insurance status or income level. The technological evolution continues accelerating with improvements in accuracy, wear time, integration capabilities, and user experience, promising even better options for future diabetes management while current systems already deliver transformative benefits for millions of users worldwide.
Frequently Asked Questions
Question 1: Are non-invasive glucose monitors FDA approved in 2026?
Answer 1: As of January 2026, the United States Food and Drug Administration has not approved any truly non-invasive glucose monitoring devices capable of measuring blood sugar levels without piercing the skin. The FDA issued a safety communication in February 2024 explicitly warning consumers against smartwatches or smart rings claiming to measure blood glucose without skin penetration, emphasizing that no such devices have received regulatory clearance or approval. Currently approved continuous glucose monitors still require sensor insertion beneath the skin surface, though they’ve become far less invasive than traditional finger-prick testing. The Dexcom Stelo represents the first over-the-counter CGM available without prescription for non-insulin users and wellness purposes, but it still uses a sensor inserted under the skin. Several companies continue developing optical and radio frequency-based technologies attempting to achieve truly non-invasive measurement, but none have demonstrated the accuracy and reliability necessary for FDA approval. Healthcare providers strongly advise against using unapproved devices for diabetes management decisions, as inaccurate readings could lead to dangerous insulin dosing errors causing severe hypoglycemia or inadequate treatment resulting in dangerous hyperglycemia.
Question 2: Can smartwatches accurately measure blood glucose levels?
Answer 2: Current commercially available smartwatches cannot independently and accurately measure blood glucose levels without additional hardware from FDA-approved continuous glucose monitors. While Apple, Samsung, Huawei, and numerous startups have invested heavily in developing non-invasive glucose sensing technologies, no company has successfully brought a device to market that meets regulatory accuracy standards for diabetes management. The technical challenges stem from the extremely weak glucose absorption signals that must be detected through multiple layers of skin tissue with varying properties, creating accuracy problems related to skin color, temperature, hydration status, and individual metabolic variations. Some smartwatches can display glucose data transmitted from separate FDA-approved CGM sensors via Bluetooth connection, providing genuine convenience value for users already wearing approved monitoring devices. This integration allows viewing real-time glucose levels and trend arrows on the wrist without requiring phone access, but the smartwatch performs only display and notification functions while the separate sensor does all actual glucose measurement. Medical organizations including the American Diabetes Association emphasize that people should never rely on unapproved smartwatch glucose claims for insulin dosing decisions, as the potentially dangerous inaccuracies could cause life-threatening treatment errors.
Question 3: What are the main differences between glucose patches and implants?
Answer 3: Glucose monitoring patches and implants differ fundamentally in sensor placement, wear duration, replacement procedures, and user experience despite both providing continuous glucose readings. Patches use sensors inserted just beneath the skin surface to depths of approximately five millimeters, remaining in place for ten to fourteen days depending on the specific system before users replace them with fresh sensors. The FreeStyle Libre 3 Plus and Dexcom G7 represent current generation patch systems featuring small adhesive-secured sensors roughly the size of coins that users apply themselves using spring-loaded applicators. Implantable systems like the Eversense 365 involve surgical placement of sensors deeper in subcutaneous tissue during brief in-office procedures performed by healthcare professionals, with sensors remaining implanted for up to one year. The implant itself stays beneath the skin continuously while users attach and remove external transmitters as needed for activities or charging, whereas patch systems remain completely external throughout their wear period. Cost structures differ substantially, with patches requiring twenty-six to fifty-two sensor replacements annually totaling approximately three thousand to six thousand dollars at retail pricing, while implants involve higher initial procedure costs but potentially lower total annual expenses due to reduced sensor replacement frequency. Insurance coverage patterns show patches receiving broader approval across more plans with simpler authorization processes, while implant coverage may require additional documentation or prior authorization demonstrating medical necessity for the surgical approach.
Question 4: How accurate are continuous glucose monitors compared to finger-prick tests?
Answer 4: Modern continuous glucose monitors achieve accuracy comparable to or exceeding traditional finger-prick blood glucose meters, with Mean Absolute Relative Difference values around eight to ten percent for leading systems. The Dexcom G7 demonstrates MARD below eight percent in clinical trials, actually surpassing many finger-stick meters that typically achieve nine to twelve percent accuracy. However, direct comparison proves complex because CGMs measure glucose in interstitial fluid rather than blood directly, creating a five to ten minute physiological lag time as glucose diffuses from blood vessels into the fluid surrounding cells. During periods of stable glucose levels, this lag causes minimal discrepancy and CGM readings closely match blood measurements. When glucose rises or falls rapidly such as after meals or during hypoglycemia treatment, the lag means interstitial readings reflect slightly outdated information compared to actual current blood glucose. The FDA has approved current generation systems as integrated CGMs meeting stringent performance criteria allowing their use in automated insulin delivery systems without confirmatory finger-sticks, reflecting confidence in their reliability for critical treatment decisions. The American Diabetes Association’s 2026 Standards of Care recommend CGM use at diabetes onset for anyone who could benefit, acknowledging the superior clinical utility of continuous readings versus intermittent finger-sticks. Healthcare providers still recommend occasional finger-stick confirmations when CGM readings seem inconsistent with symptoms, during the first day after sensor insertion, or when making major therapy changes requiring additional verification.
Question 5: Which type of glucose monitor is best for Type 1 diabetes?
Answer 5: The American Diabetes Association’s 2026 Standards of Care explicitly recommend automated insulin delivery systems as the preferred insulin delivery method for people with Type 1 diabetes, representing a significant shift from previous guidelines that treated AID as one option among many. These hybrid closed-loop systems integrate continuous glucose monitors with insulin pumps using sophisticated algorithms to automatically adjust insulin delivery based on real-time glucose data, dramatically reducing the cognitive burden of constant diabetes management while improving time in target glucose range and reducing dangerous hypoglycemic episodes. For patients not yet ready for full automation or those preferring multiple daily injections, patch-based CGM systems like Dexcom G7 or FreeStyle Libre 3 Plus paired with smart insulin pens provide substantial benefits over traditional finger-stick monitoring. Clinical studies demonstrate that Type 1 diabetes patients using CGM achieve hemoglobin A1C reductions of approximately 0.5 to 0.6 percent compared to those relying on finger-sticks alone, with greater benefits among those wearing sensors more consistently. The Standards removed previous barriers including requirements for C-peptide testing or specific diabetes duration before accessing technology, recognizing that early adoption at diagnosis provides maximum benefit by establishing good management patterns and preventing complications from the outset. Individual choices between patch and implantable systems depend on personal preferences regarding wear time, sensor replacement frequency, and tolerance for minor surgical procedures, as accuracy and clinical effectiveness remain comparable between high-quality systems within each category.
Question 6: Do insurance companies cover non-invasive glucose monitors in 2026?
Answer 6: Insurance coverage for continuous glucose monitors has expanded substantially as clinical evidence supporting their benefits has accumulated and regulatory frameworks have evolved. Most private insurance plans, Medicare, and Medicaid now cover continuous glucose monitoring for Type 1 diabetes patients with minimal copays or coinsurance, recognizing these devices as medically necessary for safe insulin management rather than optional accessories. Coverage criteria typically require documentation of diabetes diagnosis, current insulin use at specified frequency, and demonstrated engagement with diabetes self-management as evidenced by healthcare provider visits and medication adherence. Type 2 diabetes insurance coverage remains more variable, generally requiring insulin therapy with multiple daily injections, documented hypoglycemia episodes, or other specific medical indications beyond basic diabetes diagnosis. The 2026 Medicare Home Health Rule introduced significant changes to continuous glucose monitor and insulin pump coverage that raised concerns from the American Diabetes Association about potential access restrictions for beneficiaries whose current devices might not qualify under revised policies. Medicare Part B covers therapeutic CGM for beneficiaries meeting criteria including insulin administration at least three times daily, four or more finger-stick tests daily, and face-to-face physician visits every six months. State Medicaid programs show substantial variation in coverage policies, with some providing generous access comparable to private insurance while others maintain highly restrictive criteria or exclude coverage entirely, creating troubling equity issues based on geography. Over-the-counter CGM systems like Dexcom Stelo are specifically designed for consumers paying entirely out-of-pocket without insurance coverage, priced around ninety-nine dollars monthly for direct-to-consumer subscription models.
Question 7: Can glucose patches be worn while swimming or showering?
Answer 7: Current generation glucose monitoring patches are engineered as water-resistant devices specifically designed to withstand swimming, showering, and most water activities throughout their approved wear periods without compromising sensor function or adhesive integrity. The FreeStyle Libre 3 Plus, Dexcom G7, and other leading patch-based continuous glucose monitors carry water resistance ratings allowing submersion up to three feet deep for thirty minutes, making them suitable for daily hygiene routines and recreational swimming in pools or open water. The adhesive formulations balance strong bonding capable of surviving water exposure and vigorous activity against skin-friendly chemistry minimizing irritation during extended wear periods. Users report successful sensor wear during intense exercise producing significant sweating, though some athletes apply additional overlay patches to reinforce adhesion during high-impact sports or prolonged water activities. Hot tubs and saunas present more challenging conditions as elevated temperatures can affect both adhesive performance and sensor accuracy, with manufacturers generally recommending caution or temporary sensor removal during exposure to extreme heat. The transmitters attached to implantable sensors like Eversense are specifically designed as removable components, allowing users to detach the external hardware for swimming or water sports while maintaining the internal sensor function, then reattaching the transmitter afterward to resume glucose monitoring. Shower steam and humidity represent normal use conditions that quality sensors handle without difficulty, though users should verify their specific device’s water resistance rating and follow manufacturer guidelines regarding depth and duration limitations.
Question 8: What causes false readings in continuous glucose monitors?
Answer 8: Continuous glucose monitor accuracy can be compromised by numerous factors ranging from physiological responses to technical limitations and environmental conditions that users and healthcare providers must understand for safe device use. The foreign body response during the first twenty-four to forty-eight hours after sensor insertion generates inflammation and tissue changes around the sensor membrane that can temporarily degrade accuracy until the initial immune reaction subsides and tissue equilibrates. Compression of the sensor site during sleep or prolonged pressure against firm surfaces can restrict blood flow to the surrounding tissue, creating temporary inaccurate low readings that resolve once pressure releases and normal circulation resumes. Rapidly changing glucose levels during meals, exercise, or hypoglycemia treatment can cause apparent inaccuracy due to the inherent five to ten minute lag between blood glucose and interstitial fluid measurements, with the sensor reading reflecting outdated values during periods of rapid flux. Certain medications including acetaminophen interfered with older sensor models though manufacturers have largely resolved this issue in current generation devices using improved enzymatic chemistries. Dehydration affects interstitial fluid composition and glucose diffusion patterns in ways that can impact sensor performance, particularly during illness, extreme exercise, or inadequate fluid intake. Temperature extremes from either very hot or very cold environments may influence sensor electronics or tissue glucose metabolism, potentially degrading reading accuracy until normal temperature ranges restore. The FDA and device manufacturers recommend confirming unexpected readings with finger-stick blood glucose tests before making insulin dosing decisions, particularly during sensor warm-up periods, when readings seem inconsistent with symptoms, or when contemplating major therapy changes.
Question 9: How do implantable glucose sensors compare to patches for long-term use?
Answer 9: Implantable continuous glucose monitoring systems offer fundamentally different user experiences compared to patch-based alternatives, with advantages and limitations that appeal differently to various patient populations. The Eversense 365 system provides year-long continuous monitoring from a single surgically implanted sensor, eliminating the twenty-six to fifty-two sensor replacement procedures required annually with weekly or biweekly patch systems. This extended wear time appeals strongly to people frustrated by constant sensor changes, those experiencing skin irritation from adhesives requiring frequent site rotation, or individuals who simply prefer minimizing ongoing device management. The removable external transmitter allows temporary removal for contact sports, activities where external hardware proves problematic, or aesthetic reasons, while the internal sensor continues operating ready to resume data transmission when the transmitter reattaches. Clinical accuracy data shows comparable performance to leading patch systems with Mean Absolute Relative Difference values around nine percent and notably fewer false low alerts during nighttime hours, an important advantage since excessive false alarms drive many users to disable protective warnings entirely. However, implants require minor surgical procedures for both insertion and removal performed in healthcare provider offices using local anesthesia, creating barriers for people with surgical anxiety or those preferring to manage their diabetes devices independently without professional intervention. Infection risk, while extremely low with proper sterile technique, represents a theoretical concern exceeding that of patch systems where users perform sensor insertion themselves without creating surgical wounds. Insurance coverage for implants may require more extensive prior authorization demonstrating failed patch trials or specific medical justifications, and not all plans cover the Eversense system despite covering patch-based alternatives. The total cost comparison depends heavily on insurance coverage specifics, with some users finding implants more cost-effective over full years while others face higher out-of-pocket expenses compared to patch systems.
Question 10: Are there any truly non-invasive glucose monitors available without skin penetration?
Answer 10: Despite decades of intensive research investment from major technology companies, numerous well-funded startups, and academic institutions worldwide, no truly non-invasive glucose monitoring device has achieved FDA approval or demonstrated the accuracy and reliability necessary for diabetes management as of January 2026. The fundamental physics challenges preventing accurate optical glucose measurement through intact skin have proven far more difficult to overcome than early optimism suggested, with glucose representing less than one percent of skin composition and generating absorption signals easily overwhelmed by interference from water, proteins, lipids, and other compounds present in vastly higher concentrations. Technologies under active investigation include near-infrared spectroscopy, mid-infrared wavelengths showing stronger glucose absorption, Raman spectroscopy detecting molecular vibrations, radio frequency sensors measuring dielectric property changes, tear fluid analysis, saliva and sweat-based detection, and breath acetone measurement as an indirect glucose indicator. Research presentations at the 2024 MIT Workshop on Noninvasive Glucose Monitoring showcased promising laboratory results including mid-infrared sensors achieving accuracy within fifteen percent of reference measurements and Raman spectroscopy systems demonstrating glucose detection in controlled settings. However, translating these proof-of-concept demonstrations into wearable consumer devices capable of reliable performance across diverse populations and real-world conditions remains enormously challenging with no clear timeline for regulatory approval. The FDA explicitly warns consumers against purchasing smartwatches or rings claiming noninvasive glucose measurement without FDA clearance, emphasizing that these unregulated devices may provide dangerously misleading information that could cause life-threatening treatment errors. Researchers and industry observers remain cautiously optimistic that continued technological progress may eventually enable practical non-invasive monitoring for at least some applications, though breakthrough devices suitable for comprehensive diabetes management appear years rather than months away from reality.
Articles related:
Tags
📧 Get More Articles Like This
Subscribe to receive product reviews and buying guides in your inbox!
We respect your privacy. Unsubscribe at any time.